The translationally controlled tumour-associated protein (TCTP)
is a well conserved and abundantly expressed protein of the guanine
nucleotide-free chaperone family (GFC) thought to be involved in cell growth
and the allergic response.
Its function in Plasmodium falciparum,
the causal agent of cerebral malaria, remains to be validated.
While Pf-TCTP has been shown to covalently bind artemisinin2 -
a potent anti-malarial drug effective against chloroquine resistant malaria,
more recent findings suggests mechanisms of action for artemisinin
entirely independent of TCTP.3,4
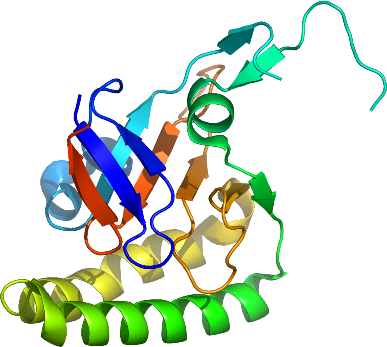
We have solved the 2.0 Å crystal structure of TCTP from the simian parasite
Plasmodium knowlesi, which has 85% identity to the P. falciparum ortholog
(PFE0545c).
Like the human TCTP structure (1YZ1), the Pk-TCTP structure is well defined except for a region
between N44 and G60.
In the NMR structure of TCTP from Schizosaccharomyces pombe (1H6Q), the same region is a highly mobile loop.
Plasmodium TCTP's include the 8 universally conserved residues of E12, S15, S46, L77, K92, E137, P157 and K170.
Of these, the triad E12, L77 and E137 are part of the putative GTPase binding surface.
Close to this surface is the mobile loop, where the conserved S46 can be found.
This serine has been reported to be a phosphorylation target.
See also