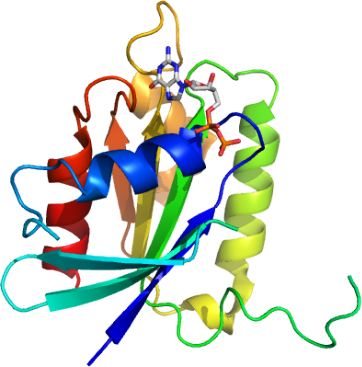
ADP-ribosylation factor-like 5 (ARL5) belongs to the ARF family of GTP-binding proteins.
The cellular function of ARL5 is currently unknown.
The primary sequence of ARL5 is most similar to that of ADP-ribosylation factor-like 8 (ARL8),
which is potentially involved in brain development.
Here, we solved the structure of the complex of ARL5 with GDP.
The ARL5-GDP structure is similar to our recent
ARL8-GDP structure (PDB code 1YZG)
as expected from their sequence similarity.
Particularly the N-terminal helix forms hydrophobic interactions with the main body of ARL5.
This N-terminal helix binding site of ARL5 is located opposite from the GTP/GDP binding site.
Based on the membrane recruitment mechanism of the ARF family proteins,
the N-terminal helix will be displaced to interact with membranes when GTP replaces GDP.
Although the ARL5-GTP structure is not known,
we hypothesize that the hydrophobic side chains of the N-terminal helix of ARL5
will be responsible for the interactions with membranes.
Further studies will be necessary to prove this hypothesis.