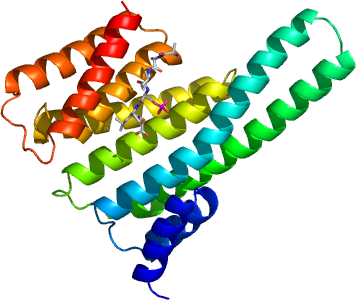
YMHAG
PDB:2B05
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:YWHAGA-s001
Entry Clone Source:MGC
SGC Clone Accession:Tag:C-terminal hexahistidine tag
Host:BL-21(DE3)R3 phage resistant
Construct
Prelude:Sequence:MVDREQLVQKARLAEQAERYDDMAAAMKN VTELNEPLSNEERNLLSVAYKNVVGARRS SWRVISSIEQKTSADGNEKKIEMVRAYRE KIEKELEAVCQDVLSLLDNYLIKNCSETQ YESKVFYLKMKGDYYRYLAEVATGEKRAT VVESSEKAYSEAHEISKEHMQPTHPIRLG LALNYSVFYYEIQNAPEQACHLAKTAFDD AIAELDTLNEDSYKDSTLIMQLLRDNLTL WTSDQQDDDGGEGNNENLYFQ
Vector:pTvHR21- SGC
Growth
Medium:Antibiotics:Procedure:Freshly transformed E. coli cells was used to inoculate 1 litre of TB plus 100 mg/ml ampicillin. When OD600 reached ~0.5 the temperature was shifted down from 37°C to 25°C for 1 hour before induction with the addition of 1 mM IPTG. Protein expression was allowed to carry on for a futher 4 hours before harvest.
Purification
ProcedureColumn 1 : Ni-affinity, HisTrap, 1 ml (GE/Amersham)
The cell extract was loaded on the column at 0.8 ml/min on an AKTA-express system (GE/Amersham). The column was then washed with 10 column volumes of WB1 and then eluted with EB at 0.8 ml/min. The eluted peak of A280 was automatically collected. Column 2 : Gel filtration, Hiload 16/60, S75 16/60 - 120 ml
The eluted fractions from the Ni-affinity Histrap columns were loaded on the gel filtration column in GF buffer at 1.0 ml/min. Eluted proteins were collected in 1 ml fractions. Using a Centricon 10 K cutoff concentrator the YWHAGA pooled fractions was concentrated to 25 mg/ml. Concentration was determined from the absorbance at 280 nm.
Enzymatic treatment : Gel filtration fractions containing YWHAGA were pooled , concentrated to 5 mg/ml using a Vivaspin 10kD cutoff spin concentrator. Next TEV protease cleavage was applied (30µl of in-house purified TEV per ml). TEV digestion was carried out at 4 ° C overnight. The following steps were carried out to remove the cleaved products and TEV protease: Place 200 µl of 50 % Ni-NTA agarose in a 1.5 ml eppendorf tubes, add 1ml of 50 mM Tris pH 8.0, 150 mM NaCl mix, spin down and remove buffer. Repeat this resin wash step once. Add the TEV treated protein sample to the resin and mix for 30 min. Finally spin down resin and collect the supernatant which contains the cleaved YWHAGA.
Extraction
ProcedureConcentration:LigandMassSpec:Before His-Tag removal
Expected MWt.: 30249.3
Measured MWt.: 30249 (main); 30118
After Hit-Tag removal
Expected MWt.: 29097.1
Measured MWt.: 29097.0
Crystallization:The purified TEV cleaved YWHAQA was then concentrated to 40 mg/ml and distributed into 30 x 50 µl aliquots and frozen at -80 ° C. First crystallisation attempts indicated that the protein concentration was too high therefore the sample was diluted to 28 mg/ml using GF buffer. YWHAGA was crystallised in the presence of a phosphorylated peptide. The peptide RSIpSLP where pS stands for a phosphorylated Serine was synthesised by Thermo Electron Corporation (www.thermo.com). The peptide was dissolved to a concentration of 40 mM in water. Before crystallisation set-up YWHAGA was mixed with the peptide to a molar ratio of 3:1 peptide-to-YWHAGA. Crystals grew from a 1:1 ratio mix of YWHAGA/peptide-to-reservoir ( 25 % PEG3350, 0.1 Bis/Tris pH 5.5, 0.2 M MgCl2).
NMR Spectroscopy:
Data Collection:Resolution: 2.55 Å; X-ray source: Synchrotron SLS -X10, single wavelength.
Data Processing: