Entry Clone Source: MGC |
Entry Clone Accession: IMAGE:3635871 |
SGC Construct ID: GMPRA-c001 |
GenBank GI number: gi|31542849 |
Vector: pLIC- SGC1. Details [PDF]; Sequence [FASTA] or [GenBank] |
Tags and additions: N-terminal His-tag with integrated TEV protease site: mgsshhhhhhssgrenlyfqghm |
Construct coding sequence:
mhhhhhhssgvdlgtenlyfqsmPRIDAD
LKLDFKDVLLRPKRSSLKSRAEVDLERTF
TFRNSKQTYSGIPIIVANMDTVGTFEMAA
VMSQHSMFTAIHKHYSLDDWKLFATNHPE
CLQNVAVSSGSGQNDLEKMTSILEAVPQV
KFICLDVANGYSEHFVEFVKLVRAKFPEH
TIMAGNVVTGEMVEELILSGADIIKVGVG
PGSVCTTRTKTGVGYPQLSAVIECADSAH
GLKGHIISDGGCTCPGDVAKAFGAGADFV
MLGGMFSGHTECAGEVIERNGRKLKLFYG
MSSDTAMNKHAGGVAEYRASEGKTVEVPY
KGDVENTILDILGGLRSTCTYVGAAKLKE
LSRRATFIRVTQQHNTVFS |
Host: E.coli BL21 Rosetta |
Growth medium, induction protocol: The GMPRA construct was expressed in E. coli (BL21 Rosetta) in 2x1 l Terrific Broth in the presence of 100 µg/ml of ampicillin and 34 µg/ml chloramphenicol at 37oC. Cells were induced at 1 mM IPTG as soon as the OD reached 0.6, and temperature was shifted to 25oC, and culture was grown for 12 hrs. Cells were collected by centrifugation, and pellets were stored frozen (-20oC) until further use. |
Extraction buffer, extraction method: Pellets were resuspended in 20 ml lysis buffer including Protease inhibitor (complete, Roche), lysed by French Press, and centrifuged to obtained a clear supernatant (15 min, 20.000 x g). Supernatants were processed in a 2 step chromatographic procedure using the Akta xpress (GE Healthcare) purification system. |
Column 1: HiTrap His |
Buffers (adjusted to pH 8.0): Lysis buffer: 10mM Imidazole, 300mM NaCl, 50mM NaH2PO4. Wash buffer: 20mM Imidazole, 300mM NaCl, 50mM NaH2PO4. Elution Buffer: 250mM Imidazole, 300mM NaCl |
Procedure: Sample was loaded, wahsed with wash buffer and eluted in elution buffer. The collected peak was injected into size-exclusion chromatography system, and the main peak was selected for concentration using an Amicon Ultra device. |
Column 2: Superdex S200 |
Buffers: 10 mM Hepes, pH 7.4, 500 mM NaCl, 5% glycerol |
Concentration: 8.5 mg/ml |
Enzymatic treatment: not performed |
Mass spec characterization: corresponds to predicted construct sequence |
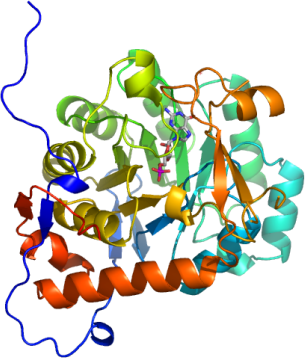
Crystallization: 2BLE: Diffraction quality crystals with overall dimensions up to 0.6*0.5*0.05 mm3 were obtained by mixing 600 nl of the protein solution (containing about 7 mM GMP) with 150 nl of the reservoir solution consisting of 0.1 M NaCacodylate pH=6.5, 15% PEG 10K, 0.10 M CaAcetate and 25% glycerol. Crystals appeared within a period of several days. 2BWG: Plate crystals with maximum dimensions 0.3*0.2*0.05 mm3 grew from drops consisting of 100 nl protein solution (containing about 5 mM GMP ) and 200 nl of 16% PEG3350 and 0.3 M K3Cit. |
Data Collection: Resolution: 2BLE: Resolution: 1.9 Å, X-ray source: SLS x10, single wavelength. 2BWG: Resolution: 2.4 Å, X-ray source: rotating anode (Rigaku FR-E SuperBright), single wavelength. |