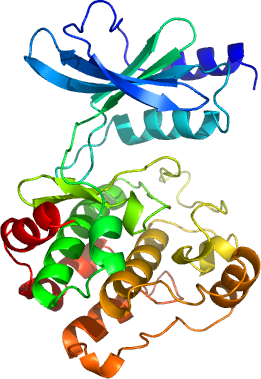
Six members of the p21-activated kinase (PAK) family of protein kinases have been identified to date. PAK's are serine/threonine kinases and can be classified into two groups named group I (PAK 1–3), and group II (PAK 4–6) based on their sequence homology and regulatory properties. In addition to the C-terminal Ste20-like catalytic domain, PAK kinases contain a Cdc42/Rac-interactive binding (CRIB) domain. PAK family members have been implicated in the regulation of multiple cellular functions, including actin reorganization, apoptotic signaling, cell motility, gene transcription, cell transformation and steroid hormone receptor signalling.
The binding of activated GTP-bound Cdc42 or Rac to group I PAKs dramatically stimulates their ability
to phosphorylate exogenous substrates.
In contrast, the group II PAK kinase PAK4 and PAK5 are active in the absence of GTPases
and their enzymatic activity is not further stimulated by binding of activated GTPase.
The reason for this difference in regulation is most likely due to the lack of an inhibitory
switch domain in group II PAK's
which releases the active site upon binding of GTPases.
However, GTPase binding has been shown to mediate kinase relocalization.
For example upon binding of Cdc42, PAK4 relocalizes to the Golgi,
and PAK5 shuttles from the microtubule network to actin-rich structures.
Recently it has been demonstrated that basal PAK6 kinase activity is repressed by a p38 mitogen-activated protein (MAP) kinase antagonist and can be stimulated by constitutively active MAP kinase kinase 6 (MKK6), an activator of p38 MAP kinases as well as by p38 MAP kinase itsself . PAK6 is directly activated by MKK6 on the activation loop residue Tyr566 once the auto-phosphorylation site Ser560 is phosphorylated. The activation of PAK6 by MKK6 and p38 suggests that PAK6 plays a role in the cellular response to stress-related signals.
PAK6 is the only PAK family member that interacts with the androgen receptor. After androgen stimulation, PAK6 binds to the ligand binding domain of the androgen receptor (AR) and translocates into the nucleus, where it inhibits AR-mediated transcription. PAK6 has also been shown to bind the estrogen receptor (ER) and to inhibit ER-mediated gene transcription.
PAK6 is mainly expressed in brain, testis, prostate, and breast tissues. No direct link to any disease has been established yet but auto-phosphorylated, active PAK6 protein is differently expressed among prostate cancer cell lines suggesting that PAK6 could be involved in differential regulation of AR signaling in these cells and possibly be involved in development and/or maintenance of androgen independence growth of tumors, which is known to be associated with aggressiveness.
See also