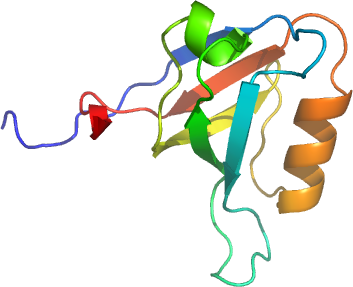
MPDZ-13
PDB:2FNE
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:MPDZA-s001
Entry Clone Source:Origene
SGC Clone Accession:Tag:N-terminal hexahistidine tag before TEV cleavage site. C-terminal PDZ recognition motif
Host:BL-21(DE3)R3 phage resistant
Construct
Prelude:Sequence:mhhhhhhssgvdlgtenlyfqsmPQCKSITLERGPDGLGFSIVGGYGSPHGDLPIYVKTVFAKGAASEDGRLKRGDQIIAVNGQSLEGVTHEEAVAILKRTKGTVTLMVLSSDETSV
Lowercase characters highlight the N-terminal hexahistidine tag and TEV cleavage recognition site.
ETSV: C-terminal extension containing PDZ recognition motif.
Vector:pNIC28-BSA4
Growth
Medium:Antibiotics:Procedure:Transformed 50 µL competent BL-21 (DE3) phage resistant cells with 10 µL of the plasmid DNA and plated out onto LB plate plus 50 µg/mL kanamycin. The next day colonies were picked out into fresh deep well blocks containing 1 mL TB + 50 µg/mL kanamycin. These were grown overnight and glycerol stocks prepared by adding 333 m l of 60 % glycerol to 1 mL of cell suspension, mixing and then storing in a -80°C freezer.
Purification
ProcedureColumn 1 : Ni-affinity, HisTrap, 1 mL (GE/Amersham)
Buffers: Affinity binding buffer: 10mM Imidazole, 300mM NaCl, 50mM pH8.0 NaH2PO4, 0.5mM TCEP; Affinity wash buffer: 50mM Imidazole, 300mM NaCl, 50mM pH8.0 NaH2PO4, 0.5mM TCEP; Affinity Elution Buffer: 250mM Imidazole, 300mM NaCl, 50mM pH8.0 NaH2PO4, 0.5mM TCEP
Procedure: The cell extract was loaded on the column at 0.8 mL/min on an AKTA-express system (GE/Amersham). The column was then washed with 10 column volumes of Affinity Binding buffer, 10 column volumes of Affinity wash buffer, and then eluted with Affinity elution buffer at 0.8 mL/min. The eluted peak of A280 was automatically collected.
Column 2 : Gel filtration, Hiload 16/60, S75 16/60 - 120 mL
Buffers : Gel Filtration: 10mM pH7.4 Hepes, 500mM NaCl, 5% glycerol, 0.5mM TCEP
Procedure: The eluted fractions from the Ni-affinity Histrap columns were loaded on the gel filtration column in GF buffer at 1.0 mL/min. Eluted proteins were collected in 1 mL fractions.
Concentration : Using a Centricon 10 K cutoff concentrator the MPDZA-p069 pooled fractions was concentrated to 7.3 mg/mL. Concentration was determined from the absorbance at 280 nm.
Extraction
ProcedureLysis buffer: 10mM Imidazole, 300mM NaCl, 50mM pH8.0 NaH2PO4, 0.5mM TCEP, 1x complete PI EDTA free tablet/50mLs. The pellet (19.52 gms) was resuspended with 3x volume of lysis buffer (approximately 50 mLs final) by intermitently placing the pellet in a 37°C water bath and vortexing. Once resuspended the cells were (1) broken by one passage through the Constant Systems cell breaker; (2) sonicating; (3) DNA precipitation with the addition of PEI to a final oncentration of 0.15 % for 30 mins on ice followed by a 17,000 rpm at 4°C to remove precipitation; (4) the supernatant was filtered through a GF/0.2 µM serum acrodiscs.
Concentration:LigandMassSpec:Crystallization:Column 1 : Ni-affinity, HisTrap, 1 mL (GE/Amersham)
Buffers: Affinity binding buffer: 10mM Imidazole, 300mM NaCl, 50mM pH8.0 NaH2PO4, 0.5mM TCEP; Affinity wash buffer: 50mM Imidazole, 300mM NaCl, 50mM pH8.0 NaH2PO4, 0.5mM TCEP; Affinity Elution Buffer: 250mM Imidazole, 300mM NaCl, 50mM pH8.0 NaH2PO4, 0.5mM TCEP
Procedure: The cell extract was loaded on the column at 0.8 mL/min on an AKTA-express system (GE/Amersham). The column was then washed with 10 column volumes of Affinity Binding buffer, 10 column volumes of Affinity wash buffer, and then eluted with Affinity elution buffer at 0.8 mL/min. The eluted peak of A280 was automatically collected.
Column 2 : Gel filtration, Hiload 16/60, S75 16/60 - 120 mL
Buffers : Gel Filtration: 10mM pH7.4 Hepes, 500mM NaCl, 5% glycerol, 0.5mM TCEP
Procedure: The eluted fractions from the Ni-affinity Histrap columns were loaded on the gel filtration column in GF buffer at 1.0 mL/min. Eluted proteins were collected in 1 mL fractions.
Concentration : Using a Centricon 10 K cutoff concentrator the MPDZA-p069 pooled fractions was concentrated to 7.3 mg/mL. Concentration was determined from the absorbance at 280 nm.
NMR Spectroscopy:
Data Collection:
Data Processing: