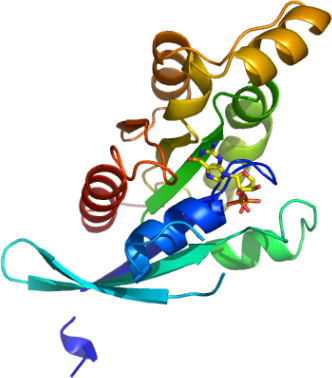
RHOB
PDB:2FV8
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:RHOBA-s001
Entry Clone Source:MGC
SGC Clone Accession:Tag:N-terminal hexahistidine tag followed by the TEV proteiease cleavable sequence
Host:BL-21(DE3)R3
Construct
Prelude:Sequence:Vector:pNIC28-Bsa4
Growth
Medium:Antibiotics:Procedure:2.5 µL of construct DNA was added to a well of a PCR plate on ice. 90µL of competent BL21(DE3)-R3/Rosetta bacteria were added, with the plate remaining on ice. The plate was left on ice for a further 25 minutes. The plate was transferred to a 42°C water bath for 45 seconds and then returned to sit on ice for 2 minutes. 100µL of SOC medium (pre-warmed to 42°C) was added and the plate incubated at 37°C for 60 minutes.180µL of the culture was plated out onto LB-agar containing 50 µg/mL kanamycin and 34 µg/mL chloramphenicol in a 5.5cm Petri dish. The plate were incubated at 37°C overnight.30mL of LB containing 50µg/mL Kanamycin and 34 µg/mL chloramphenicol was prepared in a 250 mL flask. Two colonies, from the transformation plate, were added to each starter culture. The flask was left in a shaker at 200 rpm and 37°C overnight. 19 mL of the starter culture was added to autoclaved 1litre TB medium that contained 50 µg/mL kanamycin 34 µg/mL chloramphenicol. The flasks were placed in a shaker at 180 rpm and 37°C. When the cell density reached an OD600 nm of approximately 1.0 the temperature was reduced to 20°C for 1 hr before induction with the addition of 1 mM IPTG. The cells were spun down (6500 rpm, 15 mins). The pellets were resuspended in 30 mL lysis/ binding buffer, transfered into 50 mL tubes and placed in a -80°C freezer.
Lysis buffer: 50 mM potassium phosphate pH 7.4; 10 mM Imidazole; 500 mM NaCl; 0.5 mM TCEP
Purification
ProcedureColumn 1: Ni-affinity, HisTrap, 1 mL (GE/Amersham)
Buffers: Affinity Wash Buffer: 50 mM potassium phosphate pH 7.4, 30 mM Imidazole, 500 mM NaCl, 0.5 mM TCEP
Affinity Elution Buffer: 50 mM potassium phosphate pH 7.4, 250 mM Imidazole, 500 mM NaCl, 0.5 mM TCEP
Procedure: The cell extract was loaded on the column at 0.8 mL/min on an AKTA-express system (GE/Amersham). The column was then washed with 10 column volumes of Affinity Binding buffer, 10 column volumes of Affinity wash buffer, and then eluted with Affinity elution buffer at 0.8 mL/min. The eluted peak of A280 was automatically collected.
Column 2 : Gel filtration, Hiload 16/60, S75 16/60 - 120 mL
Buffers: GF buffer: 50 mM HEPES pH 8.0, 150 mM NaCl, 2 mM MgCl 2, 0.5 mM TCEP
Procedure: The eluted fractions from the Ni-affinity Histrap columns were loaded on the gel filtration column in GF buffer at 1.0 mL/min. Eluted proteins were collected in 1 mL fractions.Using a Centricon 10 K cutoff concentrator the buffer was exchange to 10 mM HEPES pH 7.4, 500 mM NaCl, 2 mM MgCl 2 and 5 mM DTT. The volume was then brought to a total of 2 mL before adding 5 mM GDP and concetrating to concentrated to 17.9 mg/mL. Concentration was determined from the absorbance at 280 nm.
Extraction
ProcedureAfter thawing, the resuspended cells were lysed by passing through the Emusiflex C5 high pressure homogeniser. PEI (stock 5 %) was added to the homogenate to a final concentration of 0.15%. The cell debris, nuclei and DNA were spun down at 16,500 rpm for 45 min. The supernatant was collected .
Concentration:LigandMassSpec:Crystallization:Column 1: Ni-affinity, HisTrap, 1 mL (GE/Amersham)
Buffers: Affinity Wash Buffer: 50 mM potassium phosphate pH 7.4, 30 mM Imidazole, 500 mM NaCl, 0.5 mM TCEP
Affinity Elution Buffer: 50 mM potassium phosphate pH 7.4, 250 mM Imidazole, 500 mM NaCl, 0.5 mM TCEP
Procedure: The cell extract was loaded on the column at 0.8 mL/min on an AKTA-express system (GE/Amersham). The column was then washed with 10 column volumes of Affinity Binding buffer, 10 column volumes of Affinity wash buffer, and then eluted with Affinity elution buffer at 0.8 mL/min. The eluted peak of A280 was automatically collected.
Column 2 : Gel filtration, Hiload 16/60, S75 16/60 - 120 mL
Buffers: GF buffer: 50 mM HEPES pH 8.0, 150 mM NaCl, 2 mM MgCl 2, 0.5 mM TCEP
Procedure: The eluted fractions from the Ni-affinity Histrap columns were loaded on the gel filtration column in GF buffer at 1.0 mL/min. Eluted proteins were collected in 1 mL fractions.Using a Centricon 10 K cutoff concentrator the buffer was exchange to 10 mM HEPES pH 7.4, 500 mM NaCl, 2 mM MgCl 2 and 5 mM DTT. The volume was then brought to a total of 2 mL before adding 5 mM GDP and concetrating to concentrated to 17.9 mg/mL. Concentration was determined from the absorbance at 280 nm.
NMR Spectroscopy:
Data Collection:
Data Processing: