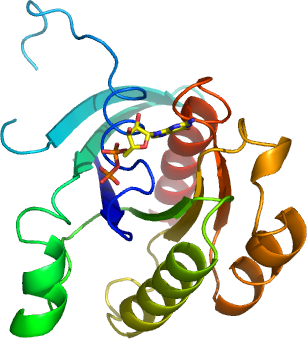
GEM
PDB:2G3Y
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:GEMA-s001
Entry Clone Source:MGC
SGC Clone Accession:Tag:mhhhhhhssgvdlgtenlyfq*s(m) TEV-cleavable (*), N-terminal his6 tag.
Host:BL-21(DE3)R3 phage resistant
Construct
Prelude:Sequence:mhhhhhhssgvdlgtenlyfqsmSTDSVI SSESGNTYYRVVLIGEQGVGKSTLANIFA GVHDSMDSDCEVLGEDTYERTLMVDGESA TIILLDMWENKGENEWLHDHCMQVGDAYL IVYSITDRASFEKASELRIQLRRARQTED IPIILVGNKSDLVRCREVSVSEGRACAVV FDCKFIETSAAVQHNVKELFEGIVRQVRL RRDSKEKN
Vector:pNIC28-Bsa4
Growth
Medium:Antibiotics:Procedure:Transformed 50 µl competent BL-21 (DE3) phage resistant cells with 10 µl of the plasmid DNA and plated out onto LB plate plus 50 µg/ml kanamycin. The next day colonies were picked out into fresh deep well blocks containing 1 ml TB + 50 µg/ml kanamycin. These were grown overnight and glycerol stocks prepared by adding 333 µl of 60 % glycerol to 1 ml of cell suspension, mixing and then storing in a -80°C freezer. The glycerol stock was used to innoculate 10 mls of TB + 50 µg/ml kanamycin which was grown overnight at 37°C as a starter culture for a 1 litre growth. The large scale growth was grown at 37°C until approximately 45 mins before induction when the temperature was lowered to 25°C. Protein production was induced with the addition of 1mM IPTG. The next day cells were harvested by centrifugation at 4000 rpm for 15 minutes. The pellet was resuspended in 50 mM K phosphate, pH 8, 500 mM NaCl, 10 mM imidazole, 0.5 mM TCEP, and then stored in the -80°C freezer.
Purification
ProcedureColumn 1: Ni Sepharose Affinity column.
The Supernatant containing the protein was loaded on to manually packed Ni sepharose beads column and washed with 30 ml of wash buffer and eluted in 6 fractions of 2 ml each using the high imidazole elute buffer. The eluate was pooled and concentrated using 10 KDa cutoff millipore filters and loaded on to Gelfiltration column.
Column 2 : Gel filtration, Hiload 16/60, S75 16/60 - 120 ml
Eluted proteins were collected in 1.75 ml fractions. Using a Centricon 10 K cutoff concentrator the buffer was exchange to 10 mM HEPES pH 7.4, 500 mM NaCl, 5% glycerol, 2 mM MgCl2 and 5 mM DTT. The volume was then brought to a total of 2 ml before adding 5 mM GDP and concentrated to 19.06 mg/ml. Concentration was determined from the absorbance at 280 nm.
Extraction
ProcedureAfter thawing, the resuspended cells were lysed by passing through Emulsiflex C5 high pressure homogeniser. PEI (stock 5 %) was added to the homogenate to a final concentration of 0.15%. The cell debris, nuclei and DNA were spun down at 16000 rpm for 45 min. The supernatant was collected.
Concentration:LigandMassSpec:Calculated mass of the full length construct is 23822.5 Da and the determined mass was 23822.7 Daltons.
Crystallization:Crystals grew from a 1:1 ratio mix of GEMA (+ GDP )-to-reservoir ( 14- 30 % PEG 3350 and 0.025 - 0.3 M Ammonium citrate pH 5.0 ).
NMR Spectroscopy:Data Collection:Resolution: 2.25 Å; X-ray source: Synchrotron ALS -8.2.1, single wavelength.
Data Processing: