The Eph family of Receptor Tyrosine Kinases is a major player in development, directing segmentation, axon guidance, fasciculation, angiogenesis, and limb development. Eph ligands, known as ephrins, are also membrane anchored, and are typically expressed on cells other than those that express receptors. Interaction between Eph- and ephrin-containing cells leads to intracellular signaling in both cell types (bi-directional signaling). What follows is a series of events that lead to either stabilization of cell-cell contact or repulsion between cells. The latter requires proteolytic degradation of the Eph receptor or ephrin ligand. Downstream signaling from Eph receptors has been associated with cell growth via the MAPK pathway, cell-cell adhesion via cadherin complexes, and cell-matrix interactions via integrin complexes.
Although not significantly present in adult tissues, Eph receptors are overexpressed during tumorigenesis. Levels of expression have been correlated with many cancers including melanoma, breast, prostate, pancreatic, gastric, esophageal and colon cancer as well as hematopoietic tumors [1-3]. Metastatic tumors also have high levels of Eph activity, consistent with the role of Eph’s in cell movement [4].
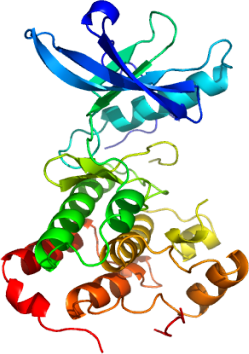
To determine the mechanism of action and regulation of the catalytic kinase domain of Epha3, and to provide a structural framework for rational drug design, we have determined its high resolution structure.
The juxtamembrane region of Eph receptors, which itself contains autophosphorylation sites, regulates catalytic activity, and although structures of related kinases suggested a mechanism whereby the juxtamembrane interferes with either inter-lobe movement or activation loop conformation [5], models of regulation remain unproven. The structure of Epha3 shows the canonical protein kinase fold and unstructured activation loop (residues 771-786).
Although completely unphosphorylated, only the first of two juxtamembrane tyrosines (Tyr596 and neighboring residues) is ordered and nestled between the amino and carboxy-terminal lobes. This conformation likely represents the inhibited form. Surprisingly, the segment of the activation loop associated with the juxtamembrane (765-770) is fully ordered and presumably in a catalytically competent conformation.
The active site of the Epha3 catalytic domain reveals features that may be unique relative to other tyrosine kinases and could be exploited for drug development. For example, Tyr701 and Thr699 are expected to interact with the adenosine moiety of ATP and provide novel points of contact with small molecule inhibitors. Further studies are underway to characterize the active site and discover new ligand scaffolds.