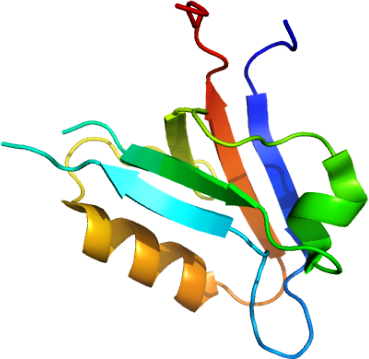
PICK1: Human protein interacting with C kinase 1 - PDZ domain
PDB:2GZV
Entry Clone Accession:PRKCABP-s001
Entry Clone Source:MGC
Host:Bl-21(DE3)R3 Phage resistant Rosetta plasmid strain
Construct
Sequence:smVPGKVTLQKDAQNLIGISIGGGAQYCP CLYIVQVFDNTPAALDGTVAAGDEITGVN GRSIKGKTKVEVAKMIQEVKGEVTIHYNK LQYYKV
Vector:pNIC28-Bsa4
Growth
Procedure:2.5 microL of construct DNA was added to a well of a PCR plate on ice. 90µl of competent BL21(DE3)-R3/Rosetta bacteria were added, with the plate remaining on ice. The plate was left on ice for a further 25 minutes. The plate was transferred to a 42°C water bath for 45 seconds and then returned to sit on ice for 2 minutes. 100µl of SOC medium (pre-warmed to 42°C) was added and the plate incubated at 37°C for 60 minutes. 180µl of the culture was plated out onto LB-agar containing 50µg/ml kanamycin and 34µg/ml chloramphenicol in a 5.5cm Petri dish. The plates were incubated at 37µC overnight. 70ml of LB containing 50µg/ml Kanamycin and 34µg/ml chloramphenicol was prepared in 250 ml flasks were prepared. A number of colonies, from the transformation plate, were added to each starter culture. The flask was left in a shaker at 180 rpm and 37°C overnight. 18 ml of the starter culture was added to autoclaved 1litre TB medium that contained 33µg/ml kanamycin. The flasks were placed in a shaker at 180 rpm and 37°C. When the cell density reached an OD
600 of approximately 1.0 the temperature was reduced to 25°C for 1 hr before induction with the addition of 1 mM IPTG. The cells were spun down (5000 rpm, 10 mins). The pellets were resuspended in 30 ml lysis/ binding buffer, transfered into 50 ml tubes and placed in a -80°C freezer.
Purification
BuffersAffinity Wash Buffer I (AWBI): 50 mM sodium phosphate pH 7.4, 500 mM NaCl, 10 mM imidazole pH 7.4, 0.5 mM TCEP
Affinity Wash Buffer II (AWBII): 50 mM sodium phosphate pH 7.4, 750 mM NaCl, 40 mM imidazole pH 7.4, 0.5 mM TCEP
Elution Buffer (EB): 50 mM sodium phosphate pH 7.4, 500 mM NaCl, 10 mM imidazole pH 7.4, 0.5 mM TCEP.
Gel Filtration: 20 mM HEPES pH 7.5, 150 mM NaCl, 0.5 mM TCEP.
ProcedureColumn 1 : Ni-affinity, HisTrap, 1 ml (GE/Amersham)
Procedure: The supernatents were filtered through a 1.2 micron and then a 0.2 micron syringe filter. Each supernatent was then diluted to a final volume of approximately 180 ml with AWBI and loaded on the HisTrap column at 0.8 ml/min on an AKTA-express system (GE/Amersham). The column was then washed with AWBI and then AWBII, both until a stable A280 trace was obtained. PRKCABPA was eluted with EB at 0.8 ml/min. The eluted peak of A280 was automatically collected.
Column 2: Gel filtration, Hiload 16/60, S75 16/60 - 120 ml
Procedure: The eluted fractions from the Ni-affinity Histrap columns were loaded on the gel filtration column in GF buffer at 1.0 ml/min. Eluted proteins were collected in 1.75 ml fractions.
Enzymatic treatment : At this stage the purity of the protein was greater than 95 % based on SDS - PAGE analysis. The C-terminal hexahistidine tag was removed by TEV protease treatment. The TEV protease, a hexahistidine-tagged construct, was over-expressed and purified in-house to a final concentration of 2.5 mg/ml. 150 µl of the TEV protease was added to the sample and left at 4°C overnight. The following steps were carried out to remove the cleaved products and TEV protease: 1.3 ml of Ni-sepharose resin was exchanged into GF buffer by repeated spinning down (350x g, 1 minute) and resuspension. 1.3 ml of the resin in GF buffer (2.6 ml of 50% slurry) was added to the sample. The sample was left rotating at 4°C for 1 hour. The resin was spun down (500x g, 3 minutes) and the supernatent filtered through a 0.2 mM syringe filter. The sample was then concentrated to 3.5 mg/ml using a 10 kD cutoff spin concentrator before storage in a -80°C freezer.
Extraction
BuffersLysis buffer: 50 mM Tris PH 7.9; 10 mM Imidazole pH 7.5; 500 mM NaCl; 5 % Glycerol; 0.5 mM TCEP.
ProcedureAfter thawing, the resuspended cells were lysed by passing through the Emusiflex C5 high pressure homogeniser. PEI (stock 5 %) was added to the homogenate to a final concentration of 0.15 %. The cell debris, nuclei and DNA were spun down at 16,500 rpm for 45 min.
MassSpec:Before His-Tag removal: Expected (MWt) 12410, Measured (MWt) 12409.8; After His-Tag removal: Expected (MWt) 9944.4, Measured (MWt) 9944.
Crystallization:Crystals grew from a 1:2 ratio mix of PRKCABPA-to-reservoir (0.2M ammonium acetate, 0.1M BIS -TRIS pH 5.5, 25 % PEG 3350 )
Data Collection:Resolution: 1.12Å X-ray source: Synchrotron SLS -X10