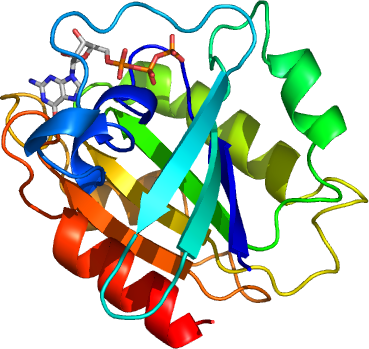
ARL6
PDB:2H57
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:BC024239
Entry Clone Source:MGC:AT30-A7
SGC Clone Accession:HPC034-B06
Tag:His-tag with integrated thrombin protease site: MGSSHHHHHHSSGLVPR*GS
Host:E. coli BL21-CodonPlus (DE-3)-RIL
Construct
Prelude:
Sequence:MGSSHHHHHHSSGLVPRGSKEVHVLCLGLDNSGKTTIINKLKPSNAQSQNILPTIGFSIEKFKSSSLSFTVFDMSGQGRYRNLWEHYYKEGQAIIFVIDSSDRLRMVVAKEELDTLLNHPDIKHRRIPILFFANKMDLRDAVTSVKVSQLLCLENIKDKPWHICASDAIKGEGLQEGVDWLQDQIQTVKT
Vector:pET28a-LIC (GI:145307000)
Growth
Medium:Terrific Broth
Antibiotics:
Procedure:We prepared the pre-growth by inoculating glycerol stock of E. coli cells BL21-CodonPlus (DE-3)-RIL into 100 mL of Luria-Bertani medium. After overnight growth, all of the pre-growth was transfered into 1.8 L of Terrific Broth medium in the presence of 50 µg/mL kanamycin and 50 µg/mL chloramphenicol at 37ºC and grown to an OD600 between 3 to 5. Cells were then induced by isopropyl-1-thio-D-galactopyranoside at the final concentration of 1.0 mM and grown overnight at 18ºC in the SGC LEX bubbling system.
Purification
Procedure
The supernatant was passed through anion exchange DE52 (Whatman) column equilibrated with the binding buffer, and then loaded onto 5 ml HisTrap HP (Amersham) equilibrated with the same binding buffer at 4ºC. The HisTrap HP column was steply washed with 25 ml of binding buffer, 25 ml of binding buffer with 30 mM imidazole, and 25 ml of binding buffer with 50 mM imidazole. The His-tagged protein was eluted by linear gradient of imidazole from 50 mM to 500 mM in 50 ml. The eluted protein peak fractions detected by UV280 nm were combined and further purified by gel filtration column Superdex 75 with a buffer containing 20 mM HEPES pH 8.0, 500m M NaCl, 1 mM DTT. Protein peak fractions were combined, GDP (Sigma) 5 times of the ARL6 in molarity, and MgCl2 to the final concentration of 5 mM MgCl2 were added before concentration. The protein was concentrated using an Amicon Ultra centrifugal filter to the final volume of 0.4 mL. The protein concentration estimated by Bradford to be 79.2 mg/mL. About 34 mg of protein was obtained from 1.8 L of cell culture.
Extraction
Procedure
Cultures were centrifuged and the cell pellets were harvested, flash frozen, and stored at -80 ºC before use. Cells were thawed and re-suspended in 100 mL the binding buffer (10 mM Tris pH 7.5, 0.5 M NaCl, 5 mM imidazole) with 0.5% CHAPS (Sigma) and 1 mM phenylmethyl sulfonyl fluoride (PMSF) and lysed with microfluidizer. The lysate was centrifuged at 15000 rpm for 30 min and the supernatant was used for subsequent steps of purification. All the extraction steps were carried out at 4ºC.
Concentration:79.2 mg/mL
Ligand
GDP supplied during crystallization, GTP-bound form crystallized.MassSpec:Expected: 21486.00, measured 21354.83.
Crystallization:Purified ARL6 was crystallized using the sitting drop vapor diffusion method at room temperature. Crystals grew in 2 days when the protein (79.2 mg/ml) was mixed with the reservoir solution in a 1:1 volume ratio, and the drop was equilibrated against a reservoir solution containing 2M (NH4)2SO4, 0.2M NaCl, 0.1 NaCaco pH5.5. The crystals were flash frozen with with the mixture of paratone-N: mineral oil at 1:1 as the cryo solution.(last updated by Y.TONG 20080220)
NMR Spectroscopy:
Data Collection:
Data Processing: