Two Plasmodium vivax 2-cys peroxiredoxin (Pv-2CP) structures have been solved: (1) oxidized Pv-2CP (2H66); and (2) reduced Pv-2CP (2I81).
As a typical 2-CysPrx, Pv-2CP is an obligate homodimer with domain swapped C-termini, such that the active site peroxidatic cysteine (Cys50-SP-) and the resolving cysteine (Cys170-SR-H) are from different subunits.
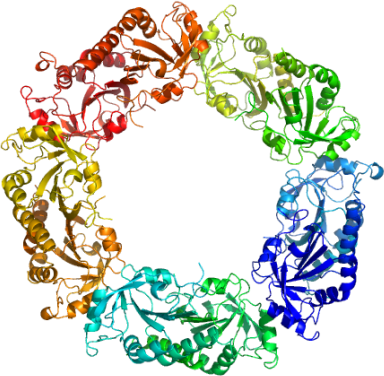
The 2-CysPrx enzyme also forms higher order oligomers (i.e. octamers, decamers and even higher).
Pv-2CP is a decamer, while
Plasmodium yoelii 2-Cys-Prx
is an octamer. The significance of these higher order oligomers is not well understood.
Peroxiredoxins catalyze the reduction of hydrogen peroxide in a two-step reaction mechanism: (1) the peroxidatic cysteine (probably as the thiolate, Cys-SP-) attacks the peroxide substrate (i.e. a hydroperoxide, peroxide, or peroxynitrite) and is oxidized to cysteinyl sulfenic acid, Cys-SP-OH, while the RO- leaving group is likely protonated; and (2) the Cys-SP-OH is subsequently attacked by the resolving cysteine (CysR) to form a stable disulfide by a condensation reaction. Then, a cellular oxidoreductase reduces the disulfide, completing the catalytic cycle. Structural information about the reduced and oxidized forms of the 2-CysPrx will aid in the understanding of this mechanism.
Reduction of a hydroperoxide to alcohol by a typical 2-CysPrx
Pv-2CP was purified in the oxidized form with at least one disulfide formed between two subunits within the homodimer.
Oxidized Pv-2CP could be reduced with 20 mM dithiothreitol or 5 mM tris(2-carboxyethyl)phosphine.
Oxidation and reduction of the enzyme was confirmed by mass spectroscopy.
The above figure shows the overall structural differences between the reduced Pv-2CP homodimer (red/pink) and the oxidized Pv-2CP homodimer (dark blue/light blue). The reduced Pv-2CP is well structured to the end of the C-terminus (residue 195), while the oxidized Pv-2CP is disordered beginning at residue 178, so the electron density could only be refined to residue 177. The figure shows this difference in the structure and length of the C-terminus. The resolving cysteine (Cys170) located at the C-terminus is in a different orientation in the reduced and oxidized forms (see below). In the oxidized structure there is a disulfide bridge formed between the two homodimers (light blue/dark blue). In the reduced structure, the C-terminal tail has an extra loop; and the two cysteines are not within disulfide forming distance or orientation.
The above electron density map shows the disulfide bridge for the oxidized form while the figure below shows no corresponding disulfide bridge for the reduced Pv-2CP.