Polyamines are essential for normal cell proliferation and cell differentiation of prokaryotes and eukaryotes.
Common polyamines such as putrescine, spermidine and spermine are distributed ubiquitously in cells and tissues at relatively high concentration.
Enzymes involved in polyamines pathway mainly include ornithine decarboxylase (ODC), S-Adenosylmethionine decarboxylase (AdoMetDC), spermidine synthase (SRM), spermine synthase (SMS), spermidine/spermine N1-acetyltransferase (SSAT) and FAD-dependent polyamine oxidase (PAO).
The polyamines are suggested to be more critical for rapidly growing and replicating P. falciparum than for normal human cells.
It has been reported that some of these polyamine enzymes exhibit features that differ significantly between the parasites and the human host.
Thus, the exploitation of such differences may lead to the design of new inhibitors that will selectively kill the parasites while exerting minimal effects on the parasite-infected host.
Spermidine synthase is an aminopropyltransferase that transfers of an aminopropyl moiety of dcAdoMet onto putrescine, leading to the formation of spermidine and 5′-methylthioadenosine (MTA).
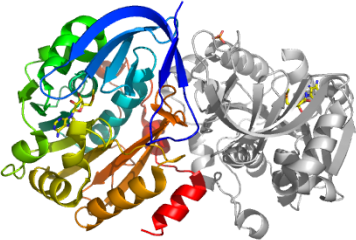
In our recently solved crystal structure of Pf-SRM, one of the products, MTA can be clearly seen in the active binding site.
Pf-SRM is a dimer in the crystal and in solution (confirmed by size exclusion chromatography).
The overall structure, as well as the substrate binding pocket is similar to that of
human SRM,
also a structure solved by the SGC. Superimposition of the two structures is presented to the right.
The Pf-SRM monomer consists of two domains: a small N-terminal domain and a large catalytic C-terminal domain (also known as SAM-dependent methyltransferase domain).
A PART (putrescine aminopropyltransferase) family signature V-(LA)-(LIV)(2)-G-G-G-X-G-X(2)-(LIV)-X-E (where X is any amino acid) sequence is also found in Pf-SRM. Some catalytic residues are located in the loop β10-αE (residue 158-169), which are disordered in two out of three molecules in the asymmetric unit.
It is of interest to note that, Pf-SRM protein was over-expressed from synthetic DNA since the gene encoding Pf-SRM consists of four exons and three introns on P. falciparum genomic DNA (www.plasmodb.org).