Calpains are calcium-activated, intracellular cyteine proteases. Some are implicated in signal transduction, amplifying or quenching signals by cleaving key intermediates. For example, proteolysis in immune cells of IKB by calpain-1, which is in turn activated by upstream calcium signaling, leads to activation of inflammatory pathways and in some cases antiapoptosis. On the other hand, injury-induced calcium influxes in neuronal tissue leads to calpain activiation, destruction of cytoskeletal, nuclear and cytosolic proteins, and subsequent cell death.
Furthermore, mutations in calpain 3 cause limb-girdle muscular dystrophies 1; wild type, active calpain 3 is necessary for proper skeletal muscle maintenance. But the exact molecular functions, including substrate specificity and subcellular localization of calpains are poorly understood.
Recent studies have shown that calpains are regulated by many different mechanisms, including calcium levels, autoproleolysis, membrane localization, and phosphorylation. Calcium activates through a complex series of events that, for many calpains, is initiated by binding to a common regulatory subunit and the regulatory domain at the Carboxy-terminus of the protein, both formed by a similar penta-EF-hand fold 2. This leads to release of constrictions to the catalytic domain and allosteric-binding of additional calcium atoms near the active site. Dysfunction of these controls are link to numerous diseases. Structures of calpains will enlighten related mechanisms and potential avenues of therapy.
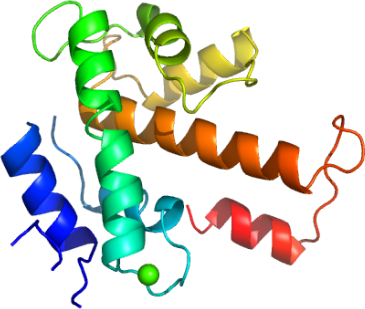
In studying human calpains, we have determined the high resolution structure of the regulatory, penta-EF-hand domain of calpain 13 (aka, C13P). C13P aligns with the analogous domain of calpain 2, whose full length structure has been determined 3, with an RMSD of 2.1 A2 (Fig. 2). As C13P was purified and crystallized in the presence of calcium, a single calcium atom is seen between α3 and α4 (Fig. 3).
Chelation occurs with the side chains of two glutamic acids (E551 and E562) and N555 (Fig. 4). Our C13P is the first calpain regulatory domain EF-hand structure in the presence of calcium, and also aligns well with the common regulatory subunit, whose structure has been determined with and without calcium 4 (Fig. 5). Many penta-EF containing proteins, including calpain 3, form dimers, and it was suggested that this interaction is mediated by the penta-EF domain 5. Although C13P does not run as a dimer according to size-exclusion chromatography, crystal contacts suggest potential for homdimerization (Fig 6).