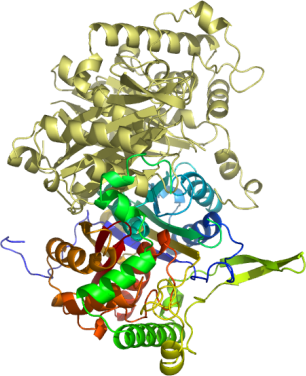
Thiolases (ketoacyl-CoA acyl transferases) are essential enzymes involved in fatty acid synthesis and catabolism, found both in pro- and eukaryotes.
In the latter, they are present in cytosolic, mitochondrial and peroxisomal subcellular compartments.
Two distinct classes (thiolase I and thiolase II) have been defined.
Thiolase I enzymes catalyze the last reaction step in the β-oxidation pathway of fatty acids resulting in one molecule each of acyl- CoA and acetyl-CoA,
which are funnelled into subsequent rounds of β-oxidation and are further oxidized.
Different forms of type I thiolases are known with distinct subcellular localization and different acyl-chain length specificities.
Human peroxisomal acetyl-CoA acyl transferase 1 (ACAA1) belongs to the type I thiolase group and acts preferentially on long- and medium-chain length acyl-CoAs.
Expression levels of ACAA1 in cultivated human hepatocytes are apparently not directly regulated by peroxisome proliferator activated-receptor alpha (PPAR a ) agonists,
although ACAA1 belongs to peroxisome proliferation-associated genes.
The human ACAA1 gene is highly expressed in liver, but also found in other metabolic highly active tissues such as CNS, muscle, heart and kidney. The human gene is localized to the short arm of chromosome 3 (3p23).
The type II thiolases are enzymes involved in fatty acid and lipid synthesis by catalyzing the condensation of two molecules of acyl-CoAs to form ketoacyl-CoA.
Despite the biological differences in these enzymes, the underlying reaction follows a reversible Claisen condensation mechanism in both cases.