Rab proteins constitute the largest branch of the Ras GTPase superfamily. Rabs use the guanine nucleotide-dependent switch mechanism common to the superfamily to regulate each of the four major steps in membrane traffic: vesicle budding, vesicle delivery, vesicle tethering, and fusion of the vesicle membrane with that of the target compartment. These different tasks are carried out by a diverse collection of effector molecules that bind to specific Rabs in their GTP-bound state.
Rab proteins distinguish themselves from other members of the Ras GTPase family with five so-called Rab family sequences F1 – F5 (F1: IGVDF; F2: KLQIW (β3); F3: RFrsiT (loop 4); F4: YYRGA (α2 – loop 5); F5: LVYDIT (β4 – loop 6)).
These sequences are located in and around switch I and switch II regions, the regions that change conformation upon GDP or GTP binding.
Rab proteins have switch conformations between GTP-Rab proteins (active form) and GDP-Rab proteins (inactive form). Effector proteins interact with the active GTP-Rab proteins. Conformational differences within the switch regions (switch I and II) of different Rab proteins lead to the specific binding of various effector proteins to the targeted Rab proteins, although effectors are also found to interact with non-conserved regions of Rab proteins, which further increasing binding specificity. In some cases, the variability in Rab-GDP switch domain structures is influenced by the presence and nature of the bound metal ion. Rab proteins share some conserved residues, these residues point their side chains at different angles in the relation to the β strands of the core. These angle shifts create very distinct surfaces of related GTPases that are surely important for their ability to interact specifically with distinct effector proteins. Thus, Rab proteins have a related overall shape, but they are very different in subtle ways that will be recognized by binding partners.
The sequence of the Rab domain of the predicted protein LOC201475 is a close homolog of Rab 12. Previous studies found that transiently expressed rab12 was localized to the Golgi complex . Rab12 was found to be associated with the atrial granules. In cultured Sertoli cells and NRK cells, rab12 was associated with small vesicle-like structures, but not with the Golgi apparatus. When overexpressed in NRK cells, rab12-associated small vesicles were not only distributed throughout the cytoplasm but also accumulated in the perinuclear cytoplasm around centrosome. All these results suggest rab 12 may function in vesicular traffic.
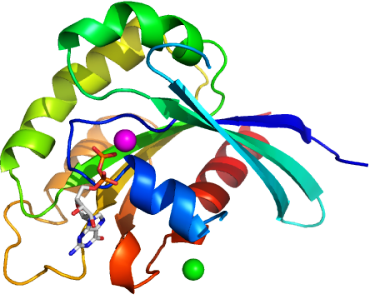
We solved the structure of the Rab domain of LOC201475 at 2.1 Å.
The overall structure of this protein shares common features with other Rab proteins with relatively subtle variations. The Rab domain of the predicted protein GDP-LOC201475 consists of a core formed by a six stranded β-sheet surrounded by five α-helices.
The conserved phosphate-binding loop (P-loop) motif in this structure is 145GXXXXGKT152, with bound Mg (II) and GDP. Superimposing this structure with crystal structure of Rab30 (2EW1) shows the conformation of switch I loop is different; this loop (T159 – S169) in the crystal structure of Rab domain of LOC201475 is disordered, the conformation of switch II (T194 – G213) remains the same as switch II of Rab 30.
Unlike Rab 30, the presence of extra metal ions may cause the disorder of switch I region. Even in conserved secondary structure regions, exposed surfaces between two proteins are different due to the different orientations of side chains. The switch regions and the some distinct surface area may play crucial roles for the potential specific binding partner recognition.