PAK-4 belongs to the STE20 subfamily of Ser/Thr protein kinases and was the first member identified of the Group II family of PAK kinases (PAK-4, PAK-5 and PAK-6). Structurally, p21-activating kinases are characterized by an N-terminal p21 binding domain (PBD). Although PBD's of the Group II PAKs are quite different from those of the Group I PAKs, they still bind to activated Rho family members. However, Cdc42 mutants like Y40C which does not bind PAK-1, 2, and 3, retains PAK-4 binding activity suggesting that the interaction of these two proteins is considerably different. In contrast to group I PAK's, binding to activated GTPases does not stimulate PAK-4 kinase activity. This difference might be explained by the lack of an identifiable inhibitory switch domain (ISD) in PAK-4 and other members of the group II family. The ISD has been shown to lock PAK-1 in an inactive state. This inhibition is released by binding of small GTPases which are therefore a key regulatory element regulating kinase activity of group I family members.
Co-expression of both PAK-4 and Cdc42 results in the translocation of PAK-4 to the golgi and stimulates the induction of filopodia in several cell types. Filopodia formation has been shown to be dependent on both PAK-4 kinases activity and its interaction with Cdc42. A recent report showed that extracellular stimuli like the cytokine HGF can stimulate PAK-4 activity and leads to the phosphorylation of the cytoskeletal regulatory protein LIMK1 and the proapoptotic protein BAD linking PAK-4 to apoptotic pathways. Indeed, activated PAK-4 has been shown to protect cells from apoptosis.
PAK-4 knockout mice are embryonic lethal by embryonic day 11.5, most likely due to a defect in the fetal heart. PAK-4 -/- embryos show neuronal abnormalities. Like neuronal differentiation has been shown to be largely inhibited, axonal outgrowth is impaired, and neurons failed to migrate to their proper locations. In addition, PAK-4 -/- embryos show defects in proper folding of the caudal portion of the neural tube, resulting in the formation of two neural lumens.
It is likely that PAK-4 plays a relevant role in oncogenic transformation. Kinase inactive PAK-4 inhibits focus formation by oncogenic Dbl or oncogenic Ras and elevated expression of PAK-4 has been reported in a high percentage of tumor cells.
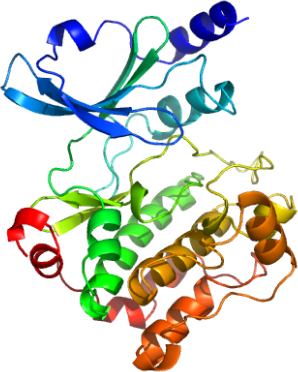
We determined therefore the structure of PAK4 in complex with the inhibitor CGP74514A; N-(cis-2-Aminocyclohexyl)-N-(3-chlorophenyl)-9-ethyl-9H-purine-2,6-diamine, which has been originally identified as a strong cdk1 kinase inhibitor. In complex with this inhibitor the entire structure of PAK4 kinase domain was well defined including the phosphorylated activation segment.
In addition, we determined two apo-structures of PAK4 using diffraction data measured on two different crystal forms. Comparison of the two high resolution structures gave interesting insights into conformational flexibility of this kinase.
We also determined the structure of PAK4 in complex with a consensus substrate peptide.