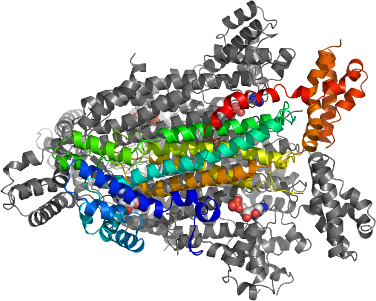
Human Adenylosuccinate Lyase (ADSL) catalyze two related reactions using one active site where reaction 1 is the final step of AMP biosynthesis.
Both of these reactions are catalysed by Adenylosuccinate Lyase:
Reaction 1: N6-(1,2-dicarboxyethyl)AMP fumarate + AMP
Reaction 2: (S)-2-[5-amino-1-(5-phospho-D-ribosyl)imidazole-4-carboxamido]succinate fumarate + 5-amino-1-(5-phospho-D-ribosyl)imidazole-4-carboxamide
We have determined the binary substrate and ternary product complexes of ADSL (reaction 1) in a single structure deposited to PDB (2VD6) and in addition the binary complex with one of its products AMP (2J91). The structure of ADSL is tetrameric and the active site is formed by three subunits of the tetramer.
When the products of reaction 1, AMP and fumarate, were soaked into our crystals the enzymatic reaction occurred “backwards†generating the covalent bond of the substrate in two of the subunits while in the other two the products AMP and fumarate remained.
Adenylosuccinate Lyase deficiency is caused by miss sense mutations in the adenylosuccinase gene. The dephosphorylated forms of the two different Adenylosuccinate Lyase substrates are accumulated in the body fluids of these patients. The clinical features of the disease are psychomotor delay and autism. The following mutations cause Adenylosuccinate Lyase deficiency: M1L, A2V, A3V, M26L, I72V, P100A, Y114H, R141W, R190Q, R194C, K246E, D268N, R303C, L311V, P318L, R337X, V364M, R374W, S395R, R396C, R396H, D422Y, L423V, R426H, D430N, S438P, S447P, T450S, R452P