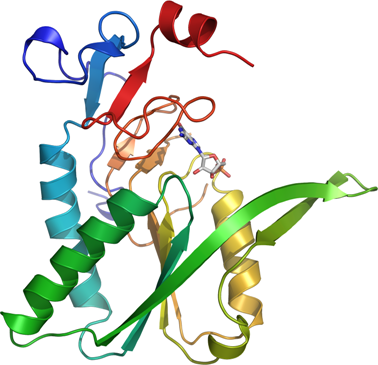
PRTFDC1A: Human phosphoribosyl transferase domain containing 1
PDB:2JBH
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:BC008662
Entry Clone Source:MGC
SGC Clone Accession:Tag:Terminal hexahistidine tag with integrated TEV protease cleavage site: mhhhhhhssgvdlgtenlyfq*s(m).
Host:Rosetta2(DE3)
Construct
Prelude:Sequence:mhhhhhhssgvdlgtenlyfqsMAGSSEEAPDYGRGVVIMDDWPGYDLNLFTYPQHYYGDLEYVLIPHGIIVDRIERLAKDIMKDIGYSDIMVLCVLKGGYKFCADLVEHLKNISRNSDRFVSMKVDFIRLKSYRNDQSMGEMQIIGGDDLSTLAGKNVLIVEDVVGTGRTMKALLSNIEKYKPNMIKVASLLVKRTSRSDGFRPDYAGFEIPNLFVVGYALDYNEYFRDLNHICVINEHGKEKYRV
Vector:pNIC28-Bsa4
Growth
Medium:Antibiotics:Procedure:BL21 (DE3) cells from glycerol stocks were grown in 100 mL LB with an addition of 50 µg/ml kanamycin at 37ºC over night. 20 mL of the over night culture inoculated 1500 mL of Terrific Broth media supplemented with 50 µg/mL kanamycin and approximately 50 µl BREOX (antifoam) in glass flasks in the Large Scale Expression System (LEX). Cells were grown at 37 ºC until OD600 of 1.2 and were down-tempered to 18 ºC for one hour in water bath. Expression of target protein was induced by addition of IPTG with a concentration of 0.5 mM. Protein expression was allowed to continue over night at 18 ºC.
Purification
ProcedureColumns: HiTrap Chelating 1 mL (IMAC); HiLoad 16/60 Superdex 200 Prep Grade (Gel filtration)
Procedure: Purification was performed on an ÄKTAprime. Prior to purification, columns were equilibrated with IMAC Bind/Wash1 Buffer (HiTrap Chelating) and Gel filtration buffer (Superdex 200). The protein sample was loaded on the HisTrap HP column that was washed with IMAC Bind/Wash1 Buffer followed by IMAC Wash2 Buffer. Bound protein was eluted from the IMAC columns with IMAC Elution Buffer and loaded in the Gel filtration column. The chromatogram from gel filtration showed one protein peak. SDS-PAGE analysis showed that the peak contained PRTFDC1A protein of high purity. Fractions corresponding to this peak were pooled and the protein was concentrated using VIVASPIN (cut off 10000) to 15 mg/mL and stored at -80 ºC.
Extraction
ProcedureCells were harvested by centrifugation and pellets were stored in -80 ºC. Prior to purification the cell pellet was resuspended in 50 mM HEPES, 500 mM NaCl, 10% glycerol, 10 mM imidazole, 0.5 mM TCEP supplemented with one tablet Complete EDTA-free protease inhibitor tablet and 4 µL/50ml benzonase. Cells were disrupted by High Pressure Homogenization run three times at 10 000 PSI and samples were centrifuged for 40 min at 20500 rpm. The soluble fraction was filtered through 0.2 µm and subjected to further purification on the ÄKTAprime.
Concentration:LigandMassSpec:Crystallization:The initial protein crystals were obtained using the JCSG screen (100 mM Sodium cacodylate pH 6.5, 200 mM calcium acetate, 40% PEG 300) co-crystallized with PRPP. In the optimized condition with 100 mM Sodium cacodylate pH 6.1, 200 mM calcium acetate and 34% PEG 300, large three dimensional crystals grew after a couple of days using hanging drop vapor diffusion. The structure was solved to 1.7Å with molecular replacement using the human hypoxanthine guanine phosphoribosyl transferase as template (1HMP).
NMR Spectroscopy:Data Collection:Data Processing: