RGS14
PDB:2JNU
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:IMAGE:4547415
Entry Clone Source:SGC Clone Accession:Tag:N-terminal, TEV cleavable (*) hexahistidine tag. Tag sequence: MHHHHHHSSGVDLGTENLYFQ(*)SM
Host:E. coli BL21(DE3)-Rosetta
Construct
Prelude:Sequence:smTEEQPVASWALSFERLLQDPLGLAYFTEFLKKEFSAENVTFWKACERFQQIPASDTQQLAQEARNIYQEFLSSQALSPVNIDRQAWLGEEVLAEPRPDMFRAQQLQIFNLMKFDSYARFVKSPLYRECLLAEAEGRPLREPGSSRLGSPDAT
Vector:pLIC-SGC1
Growth
Medium:M9
Antibiotics:Procedure:BL21(DE3)-Rosetta competent cells were transformed with the expression plasmid and plated on LB plates containing 60 µg/ml carbenicillin and 30µg/ml chloramphenicol. 4 Colonies from the transformation were used to inoculate 4 times 2 mL LB containing 60 µg/ml carbenicillin and 30 microG/mL chloramphenicol. The cells were then grown at 700 RPM, 37degC for 6 hrs. The temperature was reduced to 22degC and the cells were induced with 1 mM IPTG. Expression of the 4 clones was analyzed by SDS-PAGE. The best clone was used for large scale expression in either [
15N]-M9 or [
13C,
15N]-M9 medium, containing 0.5g [
15N]-NH
4Cl per L and, for
13C-labelling, 2g [
13C]-glucose per L.
Starter overnight cultures of 80 ml were grown at 37degC in either [15N]-M9 or [13C,15N]-M9 supplemented with 60 µg/ml carbenicillin and 30µg/ml chloramphenicol. The large scale cultures were grown in 4 x 450 ml M9 with 60 µg/ml carbenicillin and 30µg/ml chloramphenicol in 4 x 2 L bottles (dilution for inoculation 1: 25). The culture was grown at 37degC and transferred to 22degC when the OD600 reached a value of 0.5. The culture was induced with 1 mM IPTG and grown at 22degC overnight. The next day the cells were harvested by centrifugation, washed with ice-cold 150 mM NaCl and frozen at -80 degC.
Purification
ProcedureColumn 1 : Ni-affinity, MC-POROS, 8 ml (Applied Biosystems)
Procedure: The cell extract was loaded on the column at 1 ml/min on a workstation Vision (Applied Biosystems). The column was washed with 10 column volumes of start buffer and eluted with a gradient from 5 to 500 mM imidazole at a flow rate of 5 ml/min. The extinction at 280 nm was monitored and fractions were collected and analyzed by SDS-PAGE. Positive fractions were pooled and filled into a dialysis bag with 8 kDa MW cutoff.
Dialysis and TEV-cleavage: The His-tag was cleaved with 1 mg TEV per 40 mg target protein in a dialysis bag and dialysed with 5 L of: 20 mM Tris HCl 8.0, 500 mM NaCl, 1 mM ß mercaptoethanol, at 15degC overnight.
Column 2 : As column 1, but the flow rate was 0.5 ml/min and without imidazole in the start buffer.
Concentration and buffer exchange: Using Amicon Ultra-15 concentrators with 5 kDa cutoff, the RGS14A-c008 was exchanged into NMR buffer and concentrated as follows:
- U-[15N]-labelled RGS14A: final 1 mM protein in 20 mM phosphate buffer pH 6.0, 50 mM NaCl, 1mM dDTT, with 0.02% sodium azide.
- U-[13C,15N]- labelled RGS14A: protein concentration 1.7 mM in 20 mM phosphate buffer pH 6.0, 50 mM NaCl, 1mM dDTT .
Due to some impurities, the 13C,15N was re-dialysed against 2 x 5 L of 20 mM phosphate buffer pH 6.0, 50 mM NaCl, resulting in new final 13C,15N NMR sample concentration of 1.4 mM. This was later diluted to 1.3 mM and re-dialysed to a final concentration of 1.1 mM.
Samples in D2O were prepared by lyophilization and redissolving into 100% D2O.
Concentrations were determined from the absorbance at 280 nm. Procedure: The flow through was collected and concentrated.
Extraction
ProcedureFrozen cell pellets from 0.9 L culture (ca. 2.5 g wet mass) were resuspended in a total volume of 25 ml lysis buffer. The cells were broken by 2 passes at 16,000 psi through a high pressure homogeniser followed by centrifugation for 20 minutes at 60,000g. The supernatant was further clarified by filtration (0.45 µm).
Concentration:LigandMassSpec:Calculated mass of the construct was 17702 Da (
15N: 17913 Da /
13C,
15N: 18707 Da). The determined mass for the U-[15N] sample was 17891 Da and for the U-[
13C,
15N] sample was 18676 Da.
Crystallization:NMR Spectroscopy:Data Collection:Data Processing:Preliminary three dimensional structures of RGS14A were calculated using the program CYANA v. 2.0 ( Güntert et al., (1997) J. Mol. Biol. 273 , 283-298; Herrmann et al., (2002). J. Mol. Biol. 319 , 209-227 ) and were based on the resonance assignments together with NOE peak lists from
13C HMQC-NOESY, 3D
13 C-aliphatic-centred NOESY-HSQC , 3D
13 C-aromatic-centred NOESY-HSQC, and 3D 15N NOESY-HSQC spectra. Dihedral angle restraints were predicted based on the chemical shift data using the program TALOS (Cornilescu et al., J. Biomol. NMR, 13 (1999) 289-302;
http://spin.niddk.nih.gov/NMRPipe/talos/) and were used to aid initial rounds of structure calculations. Most of these were excluded in the final rounds of calculations, with the exception of a small number that helped to define a loop for which the NOE data was sparse.
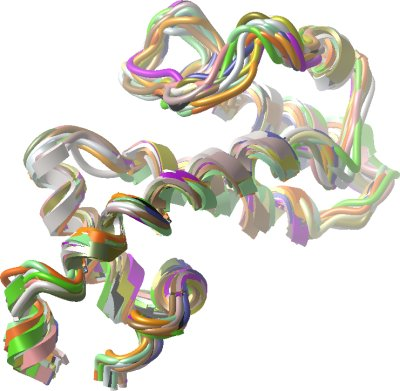
A precise ensemble of structures showing the expected RGS fold was obtained from these data. Hydrogen bond restraints were then added on the basis of the observed protected amides following sample exchange into D2O. Iterative structure refinement was carried out manually using XPLOR-NIH v. 2.14 ( Schwieters et al., (2003) J. Magn. Reson. 160 , 66-74; http://nmr.cit.nih.gov/xplor-nih/ ). Structures then underwent refinement in explicit water using CNS ( Linge et al (2003) Proteins, 50:496-506). Structures were validated using WHATIF and the Protein Structure Validation Suite (PSVS).
The refined ensemble of the 20 lowest energy structures with no NOE violations was submitted to the PDB under code 2JNU. Also included in the deposition were the chemical shift list and the list of NMR restraints used.