CTH
PDB:2NMP
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:Entry Clone Source:SGC Clone Accession:Tag:Host:Construct
Prelude:Sequence:MHHHHHHSSGVDLGTENLYFQSMQEKDASSQGFLPHFQHFATQAIHVGQDPEQWTSRAVVPPISLSTTFKQGAPGQHSGFEYSRSGNPTRNCLEKAVAALDGAKYCLAFASGLAATVTITHLLKAGDQIICMDDVYGGTNRYFRQVASEFGLKISFVDCSKIKLLEAAITPETKLVWIETPTNPTQKVIDIEGCAHIVHKHGDIILVVDNTFMSPYFQRPLALGADISMYSATKYMNGHSDVVMGLVSVNCESLHNRLRFLQNSLGAVPSPIDCYLCNRGLKTLHVRMEKHFKNGMAVAQFLESNPWVEKVIYPGLPSHPQHELVKRQCTGCTGMVTFYIKGTLQHAEIFLKNLKLFTLAESLGGFESLAELPAIMTHASVLKNDRDVLGISDTLIRLSVGLEDEEDLLEDLDQALKAAHPPSG
Vector:pNIC-Bsa4
Growth
Medium:Antibiotics:Procedure:30 microL competent BL-21 (DE3) cells were transformed with 2 µl plasmid miniprep for 30 min on ice followed by heatshock at 42°C for 45 sec. SOC, 125 µl, was added to the cellsuspension which was then incubated for 1 hour at 37°C and plated on LB-plates containing kanamycin (50 µg/mL). 20 mL TB with 100 µg kanamycin/mL was inoculated with cells and grown overnight (ON) at 30°C. The inoculation culture was added to 1.5 L TB (supplemented with 50 µg kanamycin/mL) in 2 L bottles. The flask was incubated in the LEX system-water bath at 37°C until OD600 reached 2. At this time the flask was transferred to an 18°C water bath in the LEX-system. Expression of protein was induced after approximately 1 hour by addition of 0.5 mM IPTG and continued for approximately 20 hours.Cells were harvested by centrifugation in a SLC-6000 rotor for 10 minutes at 5000 rpm (OD600 9.7; WCW 36.5 g). Pellets were suspended in 100 mL 50 mM Naphosphate pH 7.5, 10 % glycerol, 0.5 mM TCEP and 300 mM NaCl, 10mM imidazole and Complete EDTA-free protease inhibitor (Roche Biosciences). Suspended cells were stored at -80°C until further use. Before lyses, 8 µl of 250U/µl benzonase (Novagen) was added to the suspended cells and the sample was sonicated (Sonics VibraCell) at 80% amplitude for 3 min (pulse: 4Â on and 4Â off).The sample was spun for 30 min at 20500 rpm in a Sorvall SA-800 rotor. The soluble fraction was decanted and filtered through a 0.45 µm syringe filter
Purification
ProcedureIMAC and gel filtration:CTHA was purified on a Hi Trap chelating column followed by a Superdex 200 gel filtration column on the Äkta Express. The protein migrated as a tetramer on the gel-filtration column and the fractions containing protein were bright yellow in colour due to the binding of the co-factor PLP. Fractions containing protein were pooled, the TCEP-concentration adjusted to 2 mM, and concentrated to 2.7 mg/mL in amicon filters before protein aliquots were quick-frozen in liquid nitrogen.
TEV-cleavage: TEV-cleavage was performed by adding 485 µl His-tagged TEV (30 µM) to 30 mg protein in a total volume of 11.5 in gel filtration buffer with 2 mM TCEP for approximately 72 hours at 4°C. The sample was diluted with 20 mM Hepes, 500 mM NaCl, 10% glycerole and 0.5 mM TCEP at pH 7.5 before loading it onto a His-trap crude column on an Äkta Prime. The Cleaved protein bound weakly to the column and was eluted with the same buffer plus 35 mM imidazole. After elution the TCEP concentration was adjusted to 2 mM and the sample concentrated to 25.8 mg/ml in a AmiconUltra MWCO 10000 concentrator.
Extraction
ProcedureConcentration:LigandMassSpec:Crystallization:Crystals of CTHA were grown using vapor diffusion at 20°C by mixing equal amounts of protein solution at 10 mg/ml and reservoir solution containing 15% PEG3350, 200mM Ammonium Citrate pH 5.6. Plate-like crystals appeared after one day and continued to grow for one more week to reach their maximal size (approx. 80 µm × 80 µm × 10 µm).
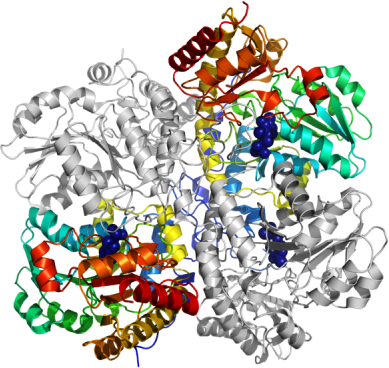
NMR Spectroscopy:Data Collection:Data Processing:The structure was solved by molecular replacement using Cystathionine gamma lyase from yeast as a search model (PDB entry: 1N8P) with the program MolRep. The asymmetric unit contains four protein monomers, making up a homotetramer. The space group was P212121 with cell dimensions a=105.78Å b=107.57Å c=153.31Å. Refmac5 was used for refinement and Coot for model building. NCS and TLS refinement using 3 TLS groups/monomer were used in the refinement process. The TLS groups were selected using the tlsmd server
http://skuld.bmsc.washington.edu/~tlsmd/. Data in the interval 19.9-2.6Å resolution was used and at the end of the refinement the values for R= 18.0% and Rfree= 24.5%. Some residues in loops are disordered and not visible in the electron density. Coordinates for the crystal structure were deposited in the Protein Data Bank, accession code 2NMP.