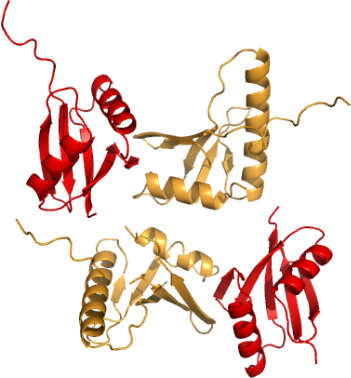
MAP2K5 + MAP3K2: Human MAP2K5 (MEK5)-MAP3K2 (MEKK2) Phox domain complex
PDB:2NPT
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:MAP2K5: NP_006600.3
MAP2K5A: NP_660143.1
Entry Clone Source:MGC
SGC Clone Accession:Tag:Tag sequence: mhhhhhhssgvdlgtenlyfq*s(m), TEV-cleavable (*) N-terminal his6 tag.
Host:BL21 (DE3)
Construct
Prelude:Sequence: MAP3K2A:
mhhhhhhssgvdlgtenlyfqSMSLQETR KAKSSSPKKQNDVRVKFEHRGEKRILQFP RPVKLEDLRSKAKIAFGQSMDLHYTNNEL VIPLTTQDDLDKAVELLDRSIHMKSLKIL LVING
MAP2K5A:
mhhhhhhssgvdlgtenlyfqSMALGPFP AMENQVLVIRIKIPNSGAVDWTVHSGPQL LFRDVLDVIGQVLPEATTTAFEYEDEDGD RITVRSDEEMKAMLSYYYSTVMEQQVNGQ LIEPLQIFPRA
Vector:pNIC28-Bsa4
Growth
Medium:Antibiotics:Procedure:Grow starter cultures from freshly transformed colonies in 10 ml LB, 50 mg/ml kanamycin. This started culture was diluted 1:1000 in fresh media and was grown at 37°C to a OD600 of 0.4 and than transferred to 18°C. Expression was induced at an OD600 of 0.6 - 0.7 using 1 mM IPTG (final concentration). Cells were harvested after 4h by centrifugation (15min, 6500rpm on a JLA 8.100 rotor), transferred to 50-ml tubes, and frozen at -20°C.
Purification
ProcedureColumn 1 : DE52/Ni-NTA
Procedure: Gravity feed chromatography. A DE52 column (10gr suspended in 100ml of 2.5M NaCl) was equilibrated with 100ml of Loading Buffer. A 5ml NiNTA column was equilibrated with 20ml of Loading Buffer. The lysed sample was applied to the DE-52 column and washed through with 50 ml loading buffer. The flow through was applied to the 5 ml Ni-NTA column which was washed with 2x10ml of wash buffer and eluted with elution buffer in 5 ml aliquots (Step elution using 50, 100, 150, 200 and 250 mM imidazole in the Elution Buffer)
Enzymatic treatment : Treated the IMAC elution(s) with TEV protease overnight.
Column 2 : SEC ( AKTA-prime)
Procedure: Gel Filtration. Each component of the complex was treated separately. Fractions containing MAP3K2 or MAP2K5 collected from IMAC and treated with TEV protease overnight (identified by SDS PAGE) were concentrated to about 1ml and directly applied to a S75 16/60 column equilibrated in 10 mM Hepes pH 7.5, 100 m NaCl. The flow rate was 1ml/min and the pure proteins eluted at 75-90min (MAP3K2) and 70-85min (MAP2K5).
Concentration : The combined samples from the SEC column (identified by SDS PAGE) were concentrated separately for each protein using centricons with 10 kDa cut off.
Column 3 : SEC (AKTA-prime)
Procedure: Gel Filtration  Complex formation. Samples of the gel-filtered proteins were mixed in 1:1 molar ratio and were directly applied to a S75 16/60 column equilibrated in 10 mM Hepes pH 7.5, 100 m NaCl. The flow rate was 1ml/min and the pure complex eluted at 60-70min.
Concentration: The combined samples from the SEC column (identified by SDS PAGE) were concentrated using centricons with 10 kDa cut off.
Extraction
ProcedureConcentration:LigandMassSpec:LC-ESI-MStof confirmed the correct masses of 11512.9Da (for MAP3K2A) and 11932.8Da (for MAP2K5A) expected for these constructs after TEV treatment.
Crystallization:Crystals were grown at 4°C in 600nl sitting drops mixing 300 nl of the complex (9 mg/ml in 10mM Hepes pH 7.5, 100mM NaCl, 10mM DTT taking into account the combined extinction coefficient) with 300 nl of a solution containing 0.2M NH4(CH3COO), 0.1M Tris pH 8.5 and 25% PEG 3350. Cryo protection was achieved by adding to the crystallization mix Ethylene Glycole (20% final w/v).
NMR Spectroscopy:Data Collection:Resolution: 1.75Å , X-ray source: Synchrotron SLS-X10, single wavelength.
Data Processing: