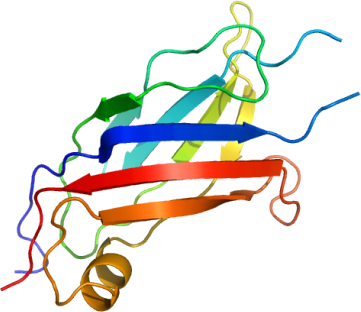
ITCH
PDB:2NQ3
Entry Clone Accession:gi:61676206; SGC clone ID: Itch.001.155, plate SDC091, well B7
Entry Clone Source:MGC
SGC Clone Accession:itch.001.155; plate SDC091:B7
Tag:mhhhhhhhssgrenlyfqg
Host:E.coli BL21 (DE3) Gold
Vector:p28a-LIC
Sequence: mhhhhhhhssgrenlyfqgMSDSGSQLGSMGSLTMKSQLQITVISAKLKENKKNWFGPSPYVEVTVDGQSKKTEKCNNTNSPKWKQPLTVIVTPVSKLHFRVWSHQTLKSDVLLGTAALDIYETLKSNNMKLEEVVVTLQLGGDKEPTETIGDLSICLDGLQLESEVVTNGETT
Growth
Procedure: The protein was expressed in E. coli BL21 (DE3) grown in Terrific Broth (TB) in the presence of 50 µg/ml of kanamycin at 37°C to an OD600 of 7.5. Cells were then induced by isopropyl-1-thio-D-galactopyranoside (IPTG), final concentration 0.1 mM, and incubated overnight at 15°C. The culture was centrifuged and the cell pellets were collected and stored at -80°C.
Purification
Procedure: IMAC purification: 4 microL of clarified supernatant is reserved for later analysis by SDS-PAGE. The rest of the clarified supernatant is then diluted 1:2 in lysis buffer, and loaded at approximately 1mL/min by gravity onto 5 mL of Ni-NTA resin (Qiagen 30450). 5 column volumes of lysis buffer are used to wash the column at approximately 3 mL/min, followed by 5 column volumes of low imidazole buffer (lysis buffer + 10 mM Imidazole (VWR EM-5720) pH 8) at approximately 3 mL/min. A 4 µL sample of the low imidazole wash is saved for later analysis by SDS-PAGE. Samples are eluted from the Ni-NTA resin by exposure to 10 mL elution buffer (lysis buffer + 250 mM imidazole and 10% glycerol (EMD GX0185-5)) at 1mL/min flow rate. A 10 µL sample of the eluate is saved for SDS-PAGE analysis. 10 µL of each eluate is saved for measurement of protein concentration using Bradford reagent (BioRad 500-0202).
Extraction
Procedure: Frozen cell pellets contained in bags (Beckman 369256) obtained from 2L liters of culture are thawed by soaking in warm water for 5 minutes. Each cell pellet is resuspended in 20 mL lysis buffer, 1mM phenylmethanesulfonyl fluoride (Sigma P7626), and 1mL Sigma general protease inhibitor (Sigma P2714-1BTL, resuspended according to manufacturer\'s instructions) and then homogenized using an Ultra-Turrax T8 homogenizer (IKA Works) at maximal setting for 30-60 seconds per pellet. Cell lysis is accomplished by sonication (Virtis408912, Virsonic) on ice: the sonication protocol is 10 sec pulse at half-maximal frequency (5.0), 10 second rest, for 6 minutes total sonication time per pellet. Lysed cells are placed into centrifuge tubes (363647, Beckman Coulter) and centrifuged in a JA25.50 rotor in an Avanti J-20 XPI centrifuge (Beckman Coulter) for 20 minutes at 69,673 x g. The supernatant is decanted into a beaker, and the insoluble pellet discarded
Structure Determination
Crystallization:Protein concentration: 20 mg/mL
Protein buffer: 20 mm TRIS-HCl, pH 8.0, 0.15 M NaCl, 5% glycerol, 2 mm DTT
Protein was mixed in 1:1 ratio with 20% PEG3350, 0.1 M BIS-TRIS, pH 6.0, 0.2 M NH4OAc, 1 mm DTT in a hanging drop plate in a temperature of 298K.
Data Collection:Resolution: 2.0Å, X-ray source: The Industrial Macromolecular Crystallography Association 17-ID beamline at the Advanced Photon Source.