Isoprenoids such as sterols and ubiquinones are a diverse and universal family of natural products. Their biosynthesis typically involves condensation of different numbers of isopentenyl diphosphate units (IPP).1 In mammals, IPP is produced by means of the mevalonate pathway with eponymous mevalonic acid as the starting metabolite. Simpler organisms, including eubacteria, algae and some plants, undergo an alternative non-mevalonate mechanism involving conversion of glyceraldehyde 3-phosphate (G3P) and pyruvate to 1-deoxy-D-xylulose 5-phosphate (DOXP) and subsequently to 2-C-methyl-D-erythritol 4-phosphate (MEP), giving rise to the DOXP pathway or the MEP pathway.
With studies uncovering the ineffectiveness of inhibitors of the mevalonate pathway, identification of enzymes such as DOXP synthase and DOXP reductoisomerase, as well as effectiveness of fosmidomycin in curing malaria in mice,2 it has been established that Plasmodium parasites produce IPP by means of the MEP pathway. Specifically, it has been shown that this pathway takes place inside the apicoplast organelle in both Plasmodium and Toxoplasma parasites.
Cryptosporidium parvum is an obligate parasite responsible for cryptosporidiosis in farm animals
and humans.
Genome sequencing and annotation has confirmed the absence of the apicoplast organelle in Cryptosporidium and apicoplast-localized pathways.
At the same time, these parasites also do not appear to have the key enzymes needed to undergo the mevalonate pathway.
Beyond this, the isoprenoid pathway in the simpler Apicomplexan organisms Cryptosporidium parvum and hominis is under-studied and remains essentially unknown.
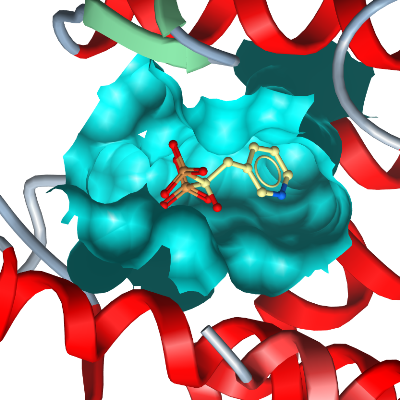
Bisphosphates are drugs that inhibit the resorption of bone.
The molecular target of nitrogen-containing bisphosphonates (N-BP) is thought to be FDPS, because these drugs have been shown to inhibit the mevalonate pathway in osteoclasts and macrophages.
In support, crystal structures of
hFDPS with N-BP have recently been solved.
The C. parvum genome encodes a gene (cgd4-2550)
which is less than 30% identical to Plasmodium and human FPPS.
We have dubbed this enzyme polyprenyl pyrophosphate synthase (PPPPS) and
have solved its structure in complex with the risedronate.
See also