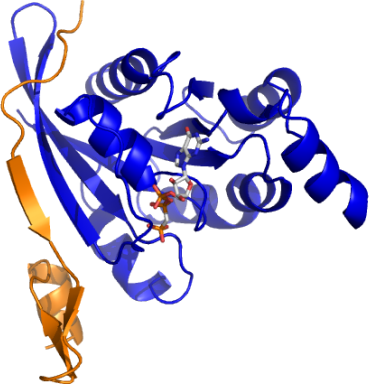
XX03CDC42A: Human Cell Division Control protein 42 in complex with human p21 activated kinase 6
PDB:2ODB
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:BC003682
Entry Clone Source:Mammalian Gene Collection
SGC Clone Accession:Tag:N-terminal, TEV cleavable hexahistidine tag
Host:BL-21(DE3)-R3-Rosetta (A homemade phage resistant version of BL21(DE3) containing the pRARE2 plasmid from Rosetta II (DE3) cells).
Construct
Prelude:Sequence: smQTIKCVVVGDGAVGKTCLLISYTTNKF PSEYVPTVFDNYAVTVMIGGEPYTLGLFD TAGQEDYDRLRPLSYPQTDVFLVCFSVVS PSSFENVKEKWVPEITHHCPKTPFLLVGT QIDLRDDPSTIEKLAKNKQKPITPETAEK LARDLKAVKYVECSALTQKGLKNVFDEAI LAALEPPEPKKSRRCVLL
Vector:pNIC28-Bsa4
Growth
Medium:TB
Antibiotics:Procedure: Transformation: The construct DNA was transformed into homemade chemically competent cells of the expression strain by a standard heat shock procedure.
Expression: A number of colonies from the transformation were used to inoculate 80 ml of LB media containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol, which was placed in a 37°C shaker overnight. The next day this starter culture was used to innoculate 4x 1L of TB medium (20 ml starter culture into each) containing 25 µg/ml kanamycin. When the OD600 reached ~1 the temperature was reduced to 25degC for 1 hour before induction with the addition of 1.0 mM IPTG. The expression was continued overnight (~ 16 hours). Cell harvest: Cells were spun at 4000 rpm for 10 mins at 4°C. Each 1L pellet was resuspended in 25 ml Resuspension Buffer. The resuspended cell pellets were placed in a -80°C freezer.
Purification
ProcedureColumn 1: IMAC. Ni-NTA (2 ml volume) in a BioRad drip column.
Procedure: 2 ml of Ni-NTA (4 ml of 50% slurry) was added to a BioRad drip column. The resin was washed with 12.5 ml of water and then 12.5 ml of WB1. The clarified cell extract was passed twice through the column. The column was then washed with 12.5 ml of WB1, and then 12.5 ml of WB2. The protein was eluted with 14 ml of EB.
Nucleotide exchange procedure and TEV protease digestion: The eluted protein was concentrated to 40 mg/ml (0.5 ml volume). To this protein solution was added EDTA (to a final concentration of 20 mM - addition of 60 µl of 0.5M stock), 100 units of Calf intestinal alkaline phosphatase (CIP), DTT (to a final concentration of 1 mM), TEV protease (addition of 100 µl of homemade protease at ~2 mg/ml) and GppCp (addition of 200 µl of 10 mM stock). The solution was left at 4°C overnight.
Column 2: Gel filtration. Hiload S200 16/60 - 120 ml volume.
Procedure: The protein solution was loaded on the gel filtration column in GF buffer at a flow rate of 1.0 ml/min. Eluted proteins were collected in 1.75 ml fractions. The fractions containing protein were identified on a coomasie blue stained gel.
Rebinding of impurities to Ni-NTA: The protein was mixed with Ni-NTA resin (pre-equilibrated into GF buffer) at 4°C for 30 minutes. The resin was spun down and the supernatent collected.
Concentration: The nucleotide exchanged and TEV protease cleaved CDC42A was concentrated to 16 mg/ml, distributed into 50 µl aliquots and frozen at -80°C.
Extraction
ProcedureConcentration:LigandMassSpec:Measured: 21345.3; Expected: 21345.5
Crystallization:Crystals grew from a 2:1 ratio of protein to precipitant solution (0.1M acetate pH 4.5; 2M (NH4)2SO2), using the vapour diffusion method.
NMR Spectroscopy:Data Collection:Crystals were cryo-protected by equilibration into precipitant solution containing 25% glycerol, and then flash frozen in liquid nitrogen. Data was collected to a resolution of 2.4 Å at the Swiss Light Source, beamline X10.
Data Processing: