|
Entry Clone Source: MGC
|
|
Entry Clone Accession: IMAGE:3349397
|
|
SGC Construct ID: CTBP2A-c009
|
|
GenBank GI number: gi|4557499
|
|
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ]
|
|
Tags and additions: N-terminal TEV-cleavable (at *) his-tag with the following sequence mhhhhhhssgvdlgtenlyfq*s.
|
Protein sequence:
mhhhhhhssgvdlgtenlyfq*sMHPRPL
VALLDGRDCTVEMPILKDLATVAFCDAQS
TQEIHEKVLNEAVGAMMYHTITLTREDLE
KFKALRVIVRIGSGYDNVDIKAAGELGIA
VCNIPSAAVEETADSTICHILNLYRRNTW
LYQALREGTRVQSVEQIREVASGAARIRG
ETLGLIGFGRTGQAVAVRAKAFGFSVIFY
DPYLQDGIERSLGVQRVYTLQDLLYQSDC
VSLHCNLNEHNHHLINDFTIKQMRQGAFL
VNAARGGLVDEKALAQALKEGRIRGAALD
VHESEPFSFAQGPLKDAPNLICTPHTAWY
SEQASLEMREAAATEIRRAITGRIPESLR
NCVNKEFFVT |
|
Host : BL21(DE3)-R3
|
|
Growth medium, induction protocol : 10ml of overnight culture was added into 1L TB supplemented with 50µg/ml of kanamycin (total 6L). The cells were cultured at 37°C until the OD reached 1.405 and then the temperature was decreased to 18°C. IPTG was added at 0.5mM (final concentration) and the culture kept at 18°C overnight.
|
|
Extraction buffer, extraction method : 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 5 mM Imidazole. Complete Protease Inhibitor Cocktail Tablets (Roche) were added (one tablet/50ml buffer). The cells were harvested by centrifugation at 4,000 g for 10 min. The pellet from 1 L culture was resuspended in 25 ml of extraction buffer. The sample was homogenized by using the EmulsiFlex-05 homogenizer (Glen Creston) and then centrifuged at 37500 g. The supernatant was kept for further purification.
|
|
Column 1 : Ni-Sepharose
|
|
Buffers: Binding buffer: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 5 mM Imidazole; Washing Buffer I: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 40 mM Imidazole; Washing Buffer II: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 50 mM Imidazole; Washing Buffer III: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 80 mM Imidazole; Elution Buffer: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 250 mM Imidazole.
|
|
Procedure: The column was packed with 4 ml of Ni-NTA slurry and equilibrated with 15 ml of binding buffer. The supernatant was loaded onto the column and the flow through was collected. The column was washed with 50 ml of binding buffer and then 20 ml of washing buffer I, 3 ml of washing buffer II & 3ml of washing buffer III. The protein was eluted with 8 ml of elution buffer and collected in 1.5 ml fractions.
|
|
Column 2 : Superdex 200 Hiload 16/60
|
|
Buffers : 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 0.5 mM TCEP.
|
|
Procedure: The gel filtration column was run using an AKTA Purifier. Fractions were collected, analyzed by SDS-PAGE gel, and pooled for further treatment.
|
| Enzymatic treatment : 300 µl of TEV protease (6mg/ml) were added into the the sample after gel filtration. The sample was incubated at 4°C overnight |
|
Column 3: Ni-Sepharose
|
|
Buffers: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 0.5 mM TCEP.
|
|
Procedure: After incubation with TEV protease, the sample was loaded onto the column (packed from 0.4 ml 0f Ni-Sepharose slurry). The flow through was collected and the column was then washed with 3 mls of the buffers with 5, 10 & 20 mM Imidazole (also collected).
|
|
Concentration : The protein was concentrated to 16.5mg/ml.
|
|
Mass spec characterization : 37229 (37228.5 expected)
|
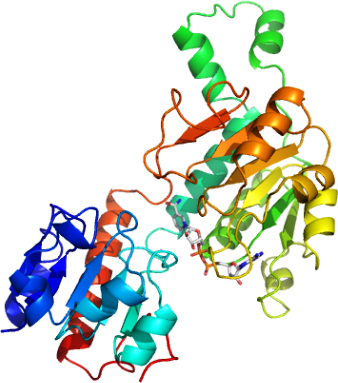
| Crystallization: Crystals were grown by vapor diffusion at 4°C in 300nl sitting drops. Prior to crystallisation NADH was added to the protein to a final concentration of 5 mM. The drops were prepared by mixing 200nl of protein solution (14mg/ml) and 100nl of precipitant consisting of 0.2M KSCN, 0.1M Bis-Tris Propane pH 6.5, 20% PEG 3350 and 10% Ethylene Glycol. Crystals were transferred to a cryo-protectant consisting of 20% Ethylene Glycol and 90% well solution before flash-cooling in liquid nitrogen. |
| Data Collection: Resolution: 2.8Å; X-ray source: Synchrotron SLS-X10, single wavelength. |