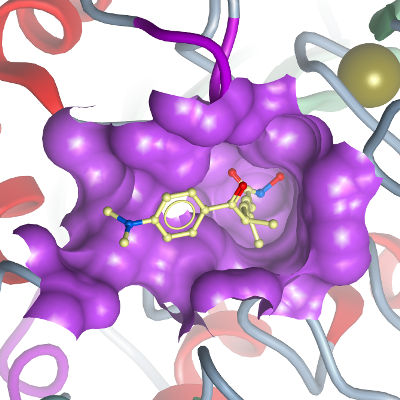
HDAC7
PDB:2PQP
Revision
Revision Type:created
Revised by:created
Revision Date:created
Entry Clone Accession:
Entry Clone Source:MGC
SGC Clone Accession:
Tag:N-terminal his tag with integrated thrombin protease site MGSSHHHHHHSSGLVPRGS
Host:E. coli BL21 (DE3) Codon Plus RIL (Stratagen)
Construct
Prelude:
Sequence:gsRAQSSPAAPASLSAPEPASQARVLSSSETPARTLPFTTGLIYDSVMLKHQCSCGDNSRHPEHAGRIQSIWSRLQERGLRSQCECLRGRKASLEELQSVHSERHVLLYGTNPLSRLKLDNGKLAGLLAQRMFVMLPCGGVGVDTDTIWNELHSSNAARWAAGSVTDLAFKVASRELKNGFAVVRPPGHHADHSTAMGFCFFNSVAIACRQLQQQSKASKILIVDWDVHHGNGTQQTFYQDPSVLYISLHRHDDGNFFPGSGAVDEVGAGSGEGFNVNVAWAGGLDPPMGDPEYLAAFRIVVMPIAREFSPDLVLVSAGFDAAEGHPAPLGGYHVSAKCFGYMTQQLMNLAGGAVVLALEGGHDLTAICDASEACVAALLGNRVDPLSEEGWKQKPNLNAIRSLEAVIRVHSKYWGCMQRLAS
Vector:p28a-LIC
Growth
Medium:
Antibiotics:
Procedure:HDAC7 was expressed in E. coli BL21 (DE3) Codon Plus RIL in Terrific Broth (TB) in the presence of 50 µg/ml of kanamycin. Cells were grown at 37 oC to an OD600 of about 2 and induced by isopropyl-1-thio-D-galactopyranoside (IPTG, final concentration 1 mM), and incubated overnight at 12 oC.
Purification
Procedure
The crude extract was cleared by centrifugation. 5 mM imidazole was added to the lysate. The sample was loaded onto 5 ml HiTrap Chelating column (Amersham Biosciences), charged with Ni2+. The column was washed with 10 CV of 20 mM Tris-HCl buffer, pH 8.0, containing 250 mM NaCl, 0.1 % CHAPS and 20 mM imidazole, and the protein was eluted with elution buffer (20 mM Tris-HCl, pH 8.0, 250 mM NaCl, 250 mM imidazole). Thrombin (Sigma) was added to combined fractions containing HDAC7 and incubated overnight at 4°C while dialyzing against 20 mM Tris-HCl, pH 8.0, 200 mM NaCl, and 5 mM ?-mercaptoethanol. The protein was further purified to homogeneity by anion-exchange chromatography on Source 30Q column (10?10) (Amersham Biosciences), equilibrated with 20 mM Tris-HCl pH 8.0 and eluted with a linear gradient of NaCl up to 400 mM (15CV). Purification yield is about 0.75 mg of protein per 1L of culture.
Extraction
Procedure
Cells were harvested by centrifugation at 7,000 rpm. The cell pellets were frozen in liquid nitrogen and stored at -80 °C. For the purification the cell paste was thawed and re-suspended in lysis buffer (1× PBS, 0.5 M NaCl, 5% glycerol, 0.1 % CHAPS, and 2.5mM ?-mercaptoethanol) with protease inhibitor (0.1 mM phenylmethyl sulfonyl fluoride, PMSF). The cells were lysed by passing through Microfluidizer (Microfluidics Corp.) at 18,000 psi.
Concentration:12 mg/mL
Ligand
MassSpec:expected mass = 45465.4 Da, measured mass = 45465.7 Da
Crystallization:2 mM NK146-I-306 (a.k.a. LAQ 824, C22H25N3O3, dissolved in DMSO, compound kindly provided from N.P. Kwiatkowski and R. Mazitschek from the Broad Institute) was directly added to concentrated protein (12 mg/mL) and incubated for 30 min on ice prior to crystallization. HDAC7 was crystallized using the hanging drop vapor diffusion method at 20 °C by mixing 2 µl of the protein solution with 2 microL of the reservoir solution containing 13 % PEG 3350, 0.1 M Hepes pH 7.5, 10 % isopropanol, and 1 mM DTT.
NMR Spectroscopy:
Data Collection:
Data Processing: