Entry clone source: MGC |
Entry clone accession : BC005252 |
Vector: pNIC28-Bsa4. Details [ PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Tags and additions: N-terminal tag: mhhhhhhssgvdlgtenlyfq |
Protein Sequence (after tag removal):
SMEKTQETVQRILLEPYKYLLQLPGKQVR
TKLSQAFNHWLKVPEDKLQIIIEVTEMLH
NASLLIDDIEDNSKLRRGFPVAHSIYGIP
SVINSANYVYFLGLEKVLTLDHPDAVKLF
TRQLLELHQGQGLDIYWRDNYTCPTEEEY
KAMVLQKTGGLFGLAVGLMQLFSDYKEDL
KPLLNTLGLFFQIRDDYANLHSKEYSENK
SFCEDLTEGKFSFPTIHAIWSRPESTQVQ
NILRQRTENIDIKKYCVHYLEDVGSFEYT
RNTLKELEAKAYKQIDARGGNPELVALVK
HLSKMFKEENE |
Host : BL21(DE3)-R3 |
Growth medium, induction protocol: Overnight cultures in TB (10 ml with50 µg/ml Kanamycin) were used to inoculate 1 litre of TB medium containing 50 µg/ml Kanamycin. Cultures were grown at 37°C until they reached an OD600 of 1-1.2 and then induced with 0.5 mM IPTG. The temperature was adjusted to 18°C and expression was allowed to continue overnight. The cells were collected by centrifugation. |
Extraction buffer, extraction method : The cell pellet was resuspended in 50 mM HEPES pH 7.5, 5 mM imidazole, 500 mM NaCl, 5% glycerol.and lysed using a high pressure cell disruptor. The lysate was centrifuged at 17,000 RPM for 30 minutes at 4°C and the supernatant was collected. |
Column 1 : 2ml Ni-NTA agarose |
Buffers: Binding: 50 mM HEPES pH 7.5, 5 mM imidazole, 500 mM NaCl, 5% glycerol, 0.5 mM TCEP; Wash: 50 mM HEPES pH 7.5, 500 mM NaCl, 30 mM imidazole, 5% glycerol, 0.5 mM TCEP ; Elution: 50 mM HEPES pH 7.5, 500 mM NaCl, 250 mM imidazole, 5% glycerol, 0.5 mM TCEP. |
Procedure: Approximately 75mls of bacterial lysate was loaded by gravity onto a 2ml Ni-NTA agarose columns pre-equilebrated with binding buffer. The columns were then washed twice with 30ml binding buffer, then twice with 12.5ml of wash buffer. Protein was then eluted with 12.5 ml of elution buffer and collected as 1.5ml fractions. Fractions containing purified protein were pooled and concentrated to a volume of less than 5mls using a Vivaspin concentrator with 10 kD MW cutoff. |
Enzymatic treatment : His-tagged TEV protease was added (25µg per mg GGPS1A) and incubated at 4°C for 48 hours to remove the hexahistidine tag. The TEV protease and any uncleaved protein was removed by passing the solution over Ni-NTA agarose beads followed by washing the beads with 15mls of wash buffer and collecting the unbound protein. The cleaved GGPS1A was then concentrated to a volume of less than 5mls using a Vivaspin concentrator with 10 kD MW cutoff. |
Column 2 : Hiload 16/60 Superdex 200 prep grade 120 ml |
Buffers : 10 mM Hepes pH 7.5, 500 mM NaCl, 5% glycerol, 0.5 mM TCEP |
Procedure: The concentrated TEV cleaved protein was loaded onto the column at 1 ml/min using an AKTA purifier system. Eluted protein was collected in 1 ml fractions. |
Concentration : The cleaved GGPS1A was concentrated to 90 mg/ml using a Vivaspin concentrator with 10 kD MW cutoff. Final protein buffer: 10 mM Hepes pH 7.5, 500 mM NaCl, 5% glycerol, 0.5 mM TCEP. |
Mass spec characterization : Characterisation of the protein by mass spectrometry revealed MW of 37.423 kDa uncleaved and 34.96kDa after TEV cleavage. |
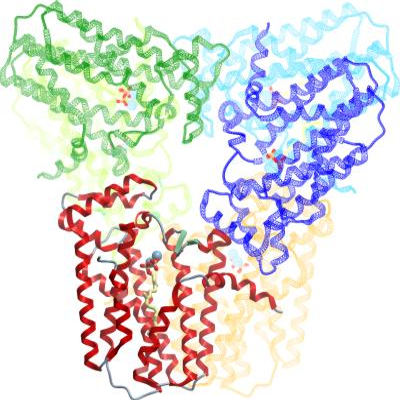
Crystallisation: Crystals were grown at 20°C in 300nl sitting drops by mixing 200nl of protein solution (90mg/ml) and 100nl of precipitant consisting of 25% PEG 3350, 0.1M Magnesium formate pH5.5. |
Data Collection: Resolution: 2.7Å; X-ray source: Synchrotron SLS -X10, single wavelength. |