Essential to all organisms, fatty acids make up all biomembranes and supply energy.
Animals typically undergo Type I fatty acid biosynthesis (FAS),
in which large multifunctional enzymes catalyze all fatty acid chain elongation reactions.
In contrast, plants and bacteria rely on the Type II system, where each elongation reaction employs a distinct enzyme.
This difference makes Type II FAS enzymes opportune targets for antibiotic drug discovery.
Protozoans, specifically apicomplexans, are mixed in their FAS mechanisms.
Malaria and Cryptosporidium parasites (C. parvum and C. hominis) respectively employ Type II and I routes,
while both mechanisms co-exist in Toxoplasma gondii.
Type II features post-translational modification of acyl carrier protein (ACP) into holo-ACP
- transfer of a 4′-phosphopantetheine (PPT) group from coenzyme A (CoA) to a conserved serine residue
by means of catalytic action of holo-ACP synthase (holo-ACPS), a type of phosphopantetheinyl transferase.
During fatty acid synthesis, the acyl intermediates are bound through a thioester linkage to this PPT moiety.
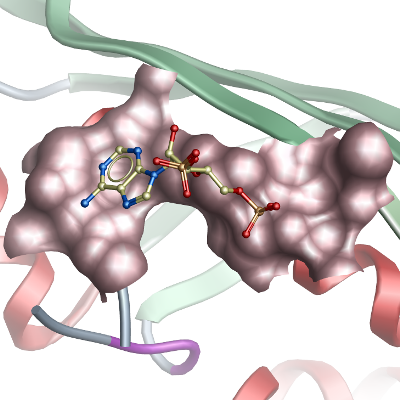
We have solved the structure of the C-terminal catalytic domain (H361-V521),
of holo-ACP synthase from Plasmodium yoelii, namely
PY06285,
that is 63% identical to the Plasmodium falciparum ortholog
PFD0980w. Here we present it as is in complex with ADP, which provides a better understanding of how substrate bound to this protein.
Py-holo-ACPS features a homo-trimer with each monomer contributing three anti-parallel beta sheets to form a barrel-like active site.
Although many FAS enzymes, including ACP, in Plasmodium genomes are targeted for the apicoplast,
this is not the case for holo-ACPS, suggesting that ACP is modified before entering the organelle.
See
other malaria related structures.