Insect and vertebrate organ formation is determined by a group of genes
that include members of the Paired box (Pax) and Homeobox (Hox) gene families.
The expression patterns of the hox gene cluster help determine cell destiny
and control the body plan of the developing embryo.
Acetylation and methylation of the hox cluster histones is required to loosen the nucleosome,
making it easier to unwind the DNA, and allowing transcription, repair, and recombination to begin.
Many of the methyl tags are known to be ferried by MLL,
a large protein containing various domains including
a histone H3 lysine 4-specific methyltransferase (SET domain).
Notably, over-expression of cyclophilin E (PPIE, CYP33),
a peptidyl prolyl isomerase that contains an amino terminal RNA recognition motif,
represses the transcription of the hox cluster and promotes deacetylation of histones
at their promoters through enhanced recruitment of HDACs.
Genetic mutations in the MLL gene has been shown to cause 10 % of all leukemias,
including acute lymphoblastic leukemia (ALL) and mixed lineage leukemia (MLL).
Many mutations are characterized by translocations and fusions with heterologous proteins,
including the E3 ligase (CBL), forkhead transcription factor (AFX1),
histone acetyltransferase (CBP), and protein phosphatase 1 (GADD34).
The chromatin structure of Hox regions is therefore likely to be involved
in the aberrant Hox gene expression patterns implicated in certain cancers.
A clearer understanding of the molecular mechanisms of MLL
and associated proteins such as PPIE may lead to better therapeutic avenues.
Post-embryonic biological roles of PPIE are not known.
Its prolyl isomerase activity could be chaperon-like,
catalyzing protein folding or reversing protien mysfolding.
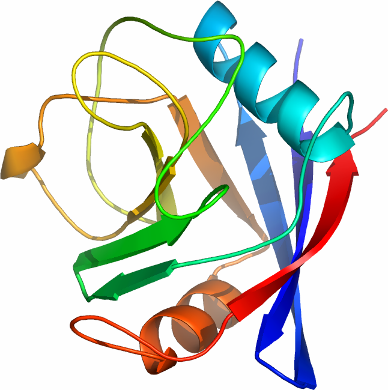
We have determined the high resolution crystal structure of isomerase domain
of human PPIE, and show that the active site contains those residues
that are important for cyclosporin binding, including F249, M197, and F196.
It enzymatic activity is expected to be intact, as the catalytic residues are conserved,
including H262 and R191.
Co-crystallization of PPIE with substrates and cyclosporin derivatives
could help in the development of novel therapeutics that target mixed lineage leukemia.