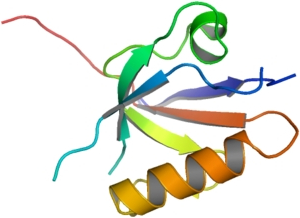
PDZ domains function as protein-protein interaction modules. The best characterised interaction of the PDZ domain is with the C-terminal 4-5 residues of the target protein that binds in an extended fashion between the betaB strand and the alphaB helix of the PDZ domain. In many cases the protein that contains the PDZ domain also possesses additional PDZ domains or interaction modules. DVL2 is one such protein which contains a Dax or DIX domain, a DEP domain, a dishevelled domain plus a PDZ domain.
The Wnt signalling pathway is important in embryogenesis and cancer. Some of the proteins involved in this signalling pathway include LDL-related protein receptor, frizzled, axin, GSK, Daam1, Rac, JNK, PKC, CamK2, beta-catenin and dishevelled. These proteins function via three independent pathways known as the canonical, non-canonical or planar cell polarity pathway and the Wnt-Ca2+ pathway. However in all three pathways a common intermediatory protein is Dishevelled (DVL).
The DVL family members interact with VANG2, a protein that when mutated results in the neural tube defect disease craniorachischisis (Torban et al, 2004). DVL2 is also part of the 'interactome' that is part of inhertied neurodegenerative disorders (Lim et al, 2006).
Here we present the crystal structure of DVL2 in complex with the C-terminal peptide -Glu-Thr-Ser-Val-COO- derived from VANGL2.