|
Entry Clone Source: MGC
|
|
Entry Clone Accession: IMAGE:5744956
|
|
SGC Construct ID: HMGCS2A-c010
|
|
GenBank GI number: gi|5031751
|
|
Vector: pNH-TrxT
|
|
Tags and additions: N-terminal Histidine/Thioredoxin -tags with TEV protease cleavage site
|
|
Expressed Sequence (Tag sequence in lower case):
mhhhhhhssgmsdkiihltddsfdtdvlk
adgailvdfwaewcgpckmiapildeiad
eyqgkltvaklnidqnpgtapkygirgip
tlllfkngevaatkvgalskgqlkeflda
nlagtenlyfqSMPKDVGILALEVYFPAQ
YVDQTDLEKYNNVEAGKYTVGLGQTRMGF
CSVQEDINSLCLTVVQRLMERIQLPWDSV
GRLEVGTETIIDKSKAVKTVLMELFQDSG
NTDIEGIDTTNACYGGTASLFNAANWMES
SSWDGRYAMVVCGDIAVYPSGNARPTGGA
GAVAMLIGPKAPLALERGLRGTHMENVYD
FYKPNLASEYPIVDGKLSIQCYLRALDRC
YTSYRKKIQNQWKQAGSDRPFTLDDLQYM
IFHTPFCKMVQKSLARLMFNDFLSASSDT
QTSLYKGLEAFGGLKLEDTYTNKDLDKAL
LKASQDMFDKKTKASLYLSTHNGNMYTSS
LYGCLASLLSHHSAQELAGSRIGAFSYGS
GLAASFFSFRVSQDAAPGSPLDKLVSSTS
DLPKRLASRKCVSPEEFTEIMNQREQFYH
KVNFSPPGDTNSLFPGTWYLERVDEQHRR
KYARRPV
|
|
Host: BL21(DE3)-R3 pRARE2
|
|
Growth medium, induction protocol: 10µl of a glycerol stock was inoculated into 5ml of TB medium (supplemented with 50µg/ml Kanamycin, 34µg/ml Chloramphenicol) and cultured at 37°C overnight in a shaking incubator (275 rpm). Next day 0.75 ml of the culture was used to inoculate 1 litre of TB medium (12 x) and grown at 37°C with vigorous shaking (160 rpm) until the culture reached an OD600 of 3.9. Temperature was reduced to 18°C, and cells were induced with IPTG at a concentration of 0.5 mM, and further cultivated for 16 hrs. Cells were harvested by centrifugation at 6500 rpm for 10 min, and the cell pellet was stored at -20°C until further use.
|
|
Extraction buffer, extraction method: Lysis buffer: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 5 mM Imidazole, Complete® protease inhibitors (Roche, 1 tbl/50 ml). Frozen cell pellets were thawed and resuspended in a total volume of 30-40 ml of lysis buffer, and disrupted by using a sonicator, and a supernatant containing the target protein was obtained by centrifugation at 21,000 (rpm) for 45 minutes.
|
|
Column 1: Ni-Sepharose 6 Fast Flow
|
|
Buffers: Lysis buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 5% Glycerol, 5 mM Imidazole; Wash buffer: 50 mM Tris-HCl pH 7.5, 500 mM NaCl, 5% Glycerol, 30 mM Imidazole; Elution buffer:50 mM HEPES pH 7.5, 500 mM NaCl, 5% Glycerol, 250 mM Imidazole. Note: All the buffers contain 0.5mM TCEP.
|
|
Procedure: The column was packed with 2 ml of Ni-Sepharose 6 Fast Flow slurry and equilibrated with 15 ml of binding buffer. The supernatant was loaded onto the column and the column was washed with 20 ml of binding buffer and then 20 ml of washing buffer. The protein was eluted with 10 ml of elution buffer.
|
|
Column 2: SuperDex 200 16/60 HiLoad (GE/Amersham)
|
|
Buffer: 10 mM HEPES, pH 7.5, 500 mM NaCl, 5 % glycerol, 0.5 mM TCEP.
|
|
Procedure: The eluted protein from the Ni-affinity column was loaded on the gel filtration column in GF buffer at 1.0 ml/min on an AKTA Purifier system. Eluted proteins were collected in 1 ml fractions.
|
|
Enzymatic treatment: TEV cleaved.
|
|
Column 3: Ni-NTA (TEV clean up)
|
|
Buffer: 10 mM HEPES, pH 7.5, 500 mM NaCl, 5% glycerol, 0.5 mM TCEP
|
|
Procedure: Total 5 mgs of protein was cleaved with 300 ug of TEV protease at 4 degree for 48 hours.
|
|
TEV clean up: The TEV cleaved protein was applied to a 1 ml Ni-NTA column, already equilibrated with gel filtration buffer (10 mM HEPES, pH 7.5, 500 mM NaCl, 5% glycerol, 0.5 mM TCEP). The flow through column was collected. The eluate from the column was monitored by SDS gel analysis.
|
|
Column 4: HP Q column (ion exchange).
|
|
PI value of protein: 6.09
|
|
Buffers: Buffer A: 20 mM Tris-Cl pH 8.5, 50mM NaCl; Buffer B: 20 mM Tris-Cl pH 8.5, 2 M NaCl.
|
|
Procedure: The target protein was applied to 5ml HP Q column in buffer A and eluted from the column by a linear gradient with buffer B.
|
|
Concentration: The target protein (in buffer; 20 mM Tris, pH 7.5, 150 mM NaCl, 0.5 mM TCEP) was concentrated to 12 mg/ml using Vivaspin 30K concentrators and stored at -80°C.
|
|
Mass spec characterization: Corresponds to theoretical mass, as determined by ESI-TOF MS.
|
|
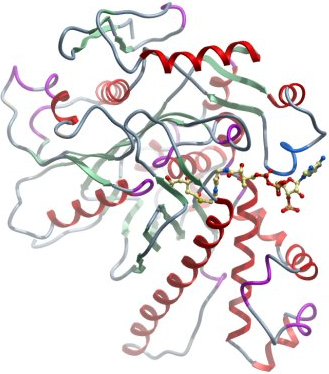
Crystallization: Crystals were grown by vapour diffusion in sitting drops at 20°C. Before setting up the experiment acetyl-CoA and acetoacetyl-CoA was added to the protein to a final concentration of 5mM. A sitting drop consisting of 100 nl protein and 50 nl well solution was equilibrated against well solution containing 0.20M ammonium sulfate, 0.1M bis-tris pH:6.5 and 25.0 %w/v polyethylene glycol 3350. Crystals were cryo protected in 25% glycerol and flash-cooled in liquid nitrogen..
|
|
Data Collection, Resolution: 1.81 Å , X-ray source: Synchrotron SLS-X10SA, single wavelength.
|