Entry Clone Source: MGC |
Entry Clone Accession: BC017723 |
SGC Construct ID: MAGEA4A-c001 |
GenBank GI number: gi|58530867 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Tags and additions: Tag sequence:mhhhhhhssgvdlgtenlyfq*s(m) TEV-cleavable (*) N-terminal his6 tag. |
Final protein sequence:
mhhhhhhssgvdlgtenlyfq*sMDAESL
FREALSNKVDELAHFLLRKYRAKELVTKA
EMLERVIKNYKRCFPVIFGKASESLKMIF
GIDVKEVDPTSNTYTLVTCLGLSYDGLLG
NNQIFPKTGLLIIVLGTIAMEGDSASEEE
IWEELGVMGVYDGREHTVYGEPRKLLTQD
WVQENYLEYRQVPGSNPARYEFLWGPRAL
AETSYVKVLEHVVRVNARVRIAYPSLREA
ALLEEEEGV |
| Host: BL21(DE3)-R3-pRARE2 |
Growth medium, induction protocol:
For native protein: The expression plasmid was transformed into the host strain and plated on LB-agar containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol. Several colonies were combined to inoculate a 1ml culture in TB (+ 50 µg/ml kanamycin, 34 µg/ml chloramphenicol). The culture was grown overnight, glycerol was added to 15% v/v (from a 60% stock), and the resulting glycerol stock was frozen at -80°C in 100 µl aliquots. A loopful of cells from the glycerol stock was inoculated into 20-ml of TB medium containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol and grown overnight at 37°C. 2x 1L TB medium (containing 50 µg/ml kanamycin) were each inoculated with 10 ml of the overnight culture and grown in 2.5L UltraYield baffled flasks until OD600 of 3.0. Cells were cooled to 18°C, IPTG added to 0.1mM and growth continued at 18°C overnight. The cells were collected by centrifugation then the pellets were scraped out and transferred to 50-ml Falcon tubes and frozen at -80°C.
For selenomethionine derivitised protein: Cells from the glycerol stock were inoculated into 6x 10-ml of LB medium containing 50 µg/ml kanamycin and 35 µg/ml chloramphenicol and grown overnight at 37°C. Cultures were harvested by centrifugation and washed twice with M9 minimal medium and resuspended in 10 ml M9 minimal medium. 6x 1L M9 minimal medium (containing 50 µg/ml kanamycin) were each inoculated with 10 ml resuspended cells and grown in 2.5L UltraYield baffled flasks until OD600 of 0.80. Selenomethioine was added to 25mg/L along with leucine, isoleucine and valine to 50mg/L and lysine, threonine, and phenylalanine to 100mg/L (all amino acids dissolved in 0.2M HEPES pH 7.5). Cultures were grown for a further 1.5 hours until OD600 of 1.2 and then cooled to 18°C for 1 hour. Additional selenomethioine was added (final total concentration of 75mg/L). IPTG was added to 0.1 mM, and growth continued at 18°C overnight. The cells were collected by centrifugation, the pellets were scraped out and transferred to 50-ml Falcon tubes and frozen at -80°C. |
Cell extraction : Lysis buffer: 50 mM Na-phosphate buffer, pH 7.5, 500 mM NaCl, 10 mM imidazole, 5% glycerol, 0.5 mM TCEP, 1x Protease Inhibitors Cocktail Set VII (Calbiochem, 1/1000 dilution), and 15 units/ml Benzonase (Novagen). 2x Lysis buffer contains the same components at double concentration. Frozen cell pellets (approx 30g) were thawed briefly in a bath of warm water (20 - 37°C) then transferred to ice. One volume (i.e. 1 ml for every gram of cells) of 2x lysis buffer was added, followed by 1x lysis buffer to a total volume of 50-ml. The cells were resuspended by agitating and disrupted by high pressure homogenization (20 kpsi). Nucleic acids and cell debris were removed by adding 0.15% PEI (polyethyleneimine) from a 5% (w/v, pH 7.5) stock, stirring for 15 minutes, then centrifugation for 20 minutes at 25,000 x g. The supernatant was then further clarified by filtration (Acrodisc filters, 0.2 µm). |
Column 1 : Ni-affinity, HisTrap Crude FF, 5 ml (GE Healthcare) |
Solutions: Affinity buffer: 50 mM Na-phosphate buffer, pH 7.5, 500 mM NaCl, 10 mM imidazole, 5% Glycerol, 0.5 mM TCEP. Wash buffer: 50 mM Na-phosphate buffer, pH 7.5, 500 mM NaCl, 20 mM imidazole, 5% Glycerol, 0.5 mM TCEP. Elution buffer: 50 mM Na-phosphate buffer, pH 7.5, 500 mM NaCl, 300 mM imidazole, 5% Glycerol, 0.5 mM TCEP. |
Procedure: The cell extract was loaded on the column at 4 ml/minute on an AKTA-express system (GE Healthcare). The column was washed with 10 volumes of lysis buffer, 10 volumes of wash buffer, and then eluted with elution buffer at 4 ml/min. The eluted peak of A280 was automatically collected. |
Column 2 : Gel filtration, Hiload 16/60 Superdex S75 prep grade, 120 ml (GE Healthcare) |
GF buffer: 10 mM HEPES, pH 7.5, 500 mM NaCl, 5% Glycerol, 0.5 mM TCEP. |
Procedure: The eluted fractions from the Ni-affinity Histrap column was loaded on the gel filtration column in GF buffer at 1.2 ml/min. Eluted proteins were collected in 2-ml fractions and analyzed on SDS-PAGE. |
Enzymatic treatment and purification: None. NOTE that the tag was not cleaved. |
Concentration method: The cleaved purified protein was concentrated in a VivaSpin4 (5 K MWCO) to 8.8 mg/ml and stored at 4°C. The protein concentration was determined spectrophotometrically using ε280 = 34380. |
Mass spectrometry characterization : Observed mass of native protein was 27381.1 (calculated mass was 27380.3 with histidine tag). Observed mass of selenomethionine-derivitised protein was 27662.6 (calculated mass was 27661.4 with selenomethionines with histidine tag). |
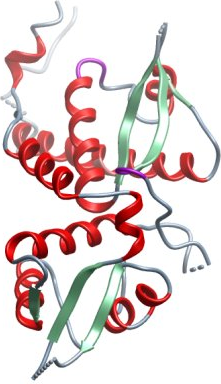
Crystallization: Crystallisation experiments were performed using the sitting drop vapour diffusion technique with drops of 75 nL protein solution and 75 nL reservoir. Native crystals of hexagonal, rod like morphology and the approximate dimensions 0.03x0.03x0.16 mm were grown at 4°C in 20% PEG3350, 10% ethylene glycol, 0.1M Bis-Tris pH 8.5 with the Emerald additive 100 mM guanidine HCl. SeMet substituted protein crystals of the same morphology were grown at 4°C in 0.3 M Na-malonate pH 7, 20% PEG 3350, 10% ethylene glycol, 0.1M Bis-Tris propane pH 8.5. Crystals were cryo protected in reservoir solution supplemented with 25% ethylene glycol and plunged into liquid nitrogen. An additional native data set was collected at Diamond light source from a larger crystal grown using the sitting drop vapour diffusion technique with drops of 100 nL protein solution and 50 nL reservoir in 2.6 M NaCl and 0.1 M Tris-HCl pH 8 and cryo protected with 20% D-glucose (w/v).
|
| Data Collection and Refinement: A two wavelength MAD data set was collected from a single SeMet crystal at the Swiss Light Source (SLS) on beamline X10SA at λ=0.9782 Å (peak) and 0.97105 Å (remote). After processing in Mosflm the data were prepared in XPREP, heavy atom sites found with SHELXD from an anomalous signal to 4 Å, and SHELXE was used to calculate the initial phases. Using SHARP the phases were extended to a first native data set (also collected at SLS beamline X10SA), which could be scaled to 2.4 Å and the maps were improved with Solomon and DM. Parts of the model could be built with buccaneer while other parts had to be built manually in Coot. Since large areas of the maps were difficult to interpret and the model wasn’t refining well, probably due to the highly anisotropic data, an additional native data set was collected at Diamond light source beamline IO4. This data improved the maps and was deposited together with the final 2.3 Å refined model. All crystals were isomorphous, belonged to the space group P65 2 2 and had an unit cell of a=82 Å, b=82 Å, c=211 Å; α=90°, =β90°, γ=120°. |