Entry Clone Source: IMAGE |
Entry Clone Accession: IMAGE:40123454 |
SGC Construct ID: ACAD11A-c306 |
GenBank GI number: gi|38505218 |
Vector: pNH-TrxT |
Amplified DNA sequence:
CATATGCACCATCATCATCATCATTCTTC
TGGTATGAGCGATAAAATTATTCACCTGA
CTGACGACAGTTTTGACACGGATGTACTC
AAAGCGGACGGGGCGATCCTCGTCGATTT
CTGGGCAGAGTGGTGCGGTCCGTGCAAAA
TGATCGCCCCGATTCTGGATGAAATCGCT
GACGAATATCAGGGCAAACTGACCGTTGC
AAAACTGAACATCGATCAAAACCCTGGCA
CTGCGCCGAAATATGGCATCCGTGGTATC
CCGACTCTGCTGCTGTTCAAAAACGGTGA
AGTGGCGGCAACCAAAGTGGGCGCACTGT
CTAAAGGTCAGTTGAAAGAGTTCCTCGAC
GCTAACCTGGCCGGTACCGAGAACTTGTA
CTTCCAATCCATGTCCAAACGAACTTTCA
GTACTGTACTACCACAGATTGATACTACT
GGACAGTTGTTTGTACAGACTCGGAAAGG
TCAGGAAGTTCTTATTAAGGTGAAGCATT
TCATGAAACAACACATTCTTCCAGCTGAA
AAGGAGGTAACTGAGTTCTATGTTCAAAA
TGAAAATTCAGTGGACAAGTGGGGAAAAC
CTTTAGTGATTGATAAACTCAAGGAAATG
GCCAAAGTCGAGGGTCTCTGGAACTTGTT
TTTGCCAGCTGTCAGCGGACTCAGCCACG
TGGACTATGCCTTGATTGCTGAAGAAACA
GGAAAATGCTTTTTTGCTCCAGATGTCTT
TAACTGCCAAGCACCAGACACAGGGAATA
TGGAGGTTCTGCACCTGTATGGAAGTGAG
GAACAGAAGAAACAGTGGCTTGAGCCTCT
TCTTCAAGGGAACATTACCTCTTGCTTCT
GTATGACAGAACCTGATGTAGCTTCAAGT
GATGCCACGAATATTGAATGCAGCATCCA
ACGAGATGAAGATAGCTATGTAATTAACG
GCAAAAAATGGTGGAGCAGTGGAGCTGGG
AATCCCAAGTGCAAAATTGCAATTGTTTT
GGGAAGAACTCAAAATACTTCTCTCTCCA
GACACAAACAGCACAGCATGATTCTTGTT
CCCATGAACACACCTGGAGTAAAAATAAT
AAGGCCTTTGTCAGTTTTTGGCTACACAG
ATAATTTTCATGGAGGACATTTTGAGATC
CATTTTAATCAAGTGCGAGTTCCTGCCAC
AAATCTAATACTAGGTGAAGGTAGGGGAT
TTGAAATTTCCCAAGGCCGCCTTGGACCT
GGCAGAATCCACCACTGTATGAGAACAGT
AGGTTTGGCGGAACGCGCTTTGCAGATCA
TGTGTGAGCGGGCAACACAAAGGATAGCT
TTCAAGAAGAAGTTGTATGCACATGAGGT
TGTGGCTCACTGGATTGCTGAAAGCCGCA
TTGCCATTGAGAAGATCCGCTTGTTGACT
CTGAAAGCTGCTCACAGCATGGACACTCT
GGGCAGTGCTGGCGCTAAGAAAGAGATTG
CAATGATCAAAGTGGCTGCCCCACGGGCT
GTCAGCAAAATCGTTGACTGGGCCATCCA
GGTGTGCGGAGGTGCTGGTGTTTCCCAGG
ATTACCCTCTGGCTAACATGTATGCTATA
ACCCGAGTTTTGCGTTTAGCAGATGGACC
TGACGAAGTTCATCTTTCAGCAATCGCAA
CAATGGAGCTGCGGGACCAAGCCAAAAGA
CTGACAGCCAAGATATAACAGTAAAGGTG
GATACGGATCCGAATTCGAGCTCCGTCGA
CAAGCTT |
Tags and additions: N terminal 6x histidines had been cleaved by TEV. N-terminal TEV-cleavable (at *) tags with the following sequence:
mhhhhhhssgmsdkiihltddsfdtdvlk
adgailvdfwaewcgpckmiapildeiad
eyqgkltvaklnidqnpgtapkygirgip
tlllfkngevaatkvgalskgqlkeflda
nlagtenlyfq*s |
Protein Construct sequence (tag residues in lowercase): |
mhhhhhhssgmsdkiihltddsfdtdvlk
adgailvdfwaewcgpckmiapildeiad
eyqgkltvaklnidqnpgtapkygirgip
tlllfkngevaatkvgalskgqlkeflda
nlagtenlyfq*sMSKRTFSTVLPQIDTT
GQLFVQTRKGQEVLIKVKHFMKQHILPAE
KEVTEFYVQNENSVDKWGKPLVIDKLKEM
AKVEGLWNLFLPAVSGLSHVDYALIAEET
GKCFFAPDVFNCQAPDTGNMEVLHLYGSE
EQKKQWLEPLLQGNITSCFCMTEPDVASS
DATNIECSIQRDEDSYVINGKKWWSSGAG
NPKCKIAIVLGRTQNTSLSRHKQHSMILV
PMNTPGVKIIRPLSVFGYTDNFHGGHFEI
HFNQVRVPATNLILGEGRGFEISQGRLGP
GRIHHCMRTVGLAERALQIMCERATQRIA
FKKKLYAHEVVAHWIAESRIAIEKIRLLT
LKAAHSMDTLGSAGAKKEIAMIKVAAPRA
VSKIVDWAIQVCGGAGVSQDYPLANMYAI
TRVLRLADGPDEVHLSAIATMELRDQAKR
LTAKI |
Host: BL21(DE3)-R3-pRARE2 |
Growth medium, induction protocol: TB. 10ml of overnight culture was added into 1L TB with 50 µg/ml of Kanamycin and 34 µg/ml of chloramphencol (totol 4L). The cells were cultured at 37°C until the OD600 reached 1.632 and then decreased the temperature to 18°C. IPTG was added at 0.1mM (final concentration) and kept the culture at 18°C for overnight. |
Extraction buffer, extraction method: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 5 mM Imidazole. The cells were harvested by centrifugation at 4,000 g for 10 min. The pellet from 1 L culture was resuspended in 25 ml of extraction buffer. The sample was homogenized by using the EmulsiFlex-05 homogenizer (Glen Creston) and then centrifuged at 37505 g. The supernatant was kept for further purification. |
Column 1: Ni-NTA |
Buffers: Binding buffer: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 5 mM Imidazole; Washing Buffer: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 30 mM Imidazole; Elution Buffer I: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 60 mM Imidazole; Elution Buffer II: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 80 mM Imidazole; Elution Buffer III: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 125 mM Imidazole; Elution Buffer VI: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 250 mM Imidazole. |
Procedure: The column was packed by 6 ml of Ni-NTA slurry and equilibrated with 15 ml of binding buffer. The supernatant was loaded onto the column and the flow through was collected. The column was washed with 2x15 ml of of washing buffer. The protein was eluted with 5 ml of elution buffer I, II & III respectively and then 12 ml of elution buffer VI. |
Column 2: Ni-NTA |
Buffers: 500 mM NaCl, 5% Glycerol, 50 mM HEPES pH 7.5, 0.5 mM TCEP. |
Procedure: The tags were cleaved by TEV protease and the buffer had been changed to the buffer without Imidazole. The sample was then loaded onto the column (packed from 2.5 ml of Ni-NTA slurry). The flow through was collected and the the column was then washed with 3 ml of the buffer (also collected). |
Concentration: 42 mg/ml |
Enzymatic treatment: 400 ml of TEV protease were added into the the sample after the first Ni-NTA purification. The sample was incubated at 4°C overnight |
Mass spectrometry characterization: 47645 |
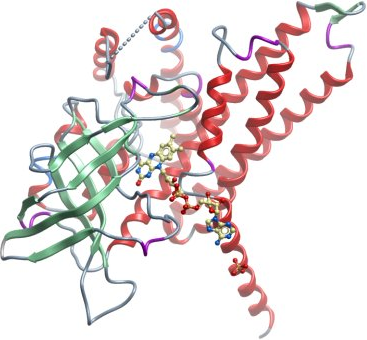
Crystallisation: Crystals were grown by vapour diffusion at 20°C in 300 nl sitting drops. FAD to a final concentration of 2 mM was added to the protein just prior to crystallisation. The drops were prepared by mixing 150 nl of protein solution and 150 nl of buffer consisting of 0.2 M NaCl, 0.1 M Na/P-PO4 pH 6.2 and 50% PEG 300. |
Data collection: Resolution: 2.8 Å. X-ray source: beamline X10SA at the Swiss light source. |