|
Entry Clone Source: MGC
|
|
Entry Clone Accession: IMAGE:6066312
|
|
SGC Construct ID: GLRX5A-c006
|
|
GenBank GI number: gi|42516576
|
|
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ]
|
|
Amplified construct sequence:
CATATGCACCATCATCATCATCATTCTTC
TGGTGTAGATCTGGGTACCGAGAACCTGT
ACTTCCAATCCATGGGCGCGGGCGGCGGC
GGCTCGGCGGAGCAGTTGGACGCGCTGGT
GAAGAAGGACAAGGTGGTGGTCTTCCTCA
AGGGGACGCCGGAGCAGCCCCAGTGCGGC
TTCAGCAACGCCGTGGTGCAGATCCTGCG
GCTGCACGGCGTCCGCGATTACGCGGCCT
ACAACGTGCTGGACGACCCGGAGCTCCGA
CAAGGCATTAAAGACTATTCCAACTGGCC
CACCATCCCGCAAGTGTACCTCAATGGCG
AGTTTGTAGGGGGCTGTGACATTCTTCTG
CAGATGCACCAGAATGGGGACTTGGTGGA
AGAACTGAAAAAGCTGGGGATCCACTCCG
CCCTTTTAGATGAATGACAGTAAAGGTGG
ATACGGATCCGAA
|
|
Tags and additions: TEV-cleavable (*), N-terminal histag. Tag sequence: mhhhhhhssgvdlgtenlyfq*sm
|
|
Protein sequence (after TEV cleavage):
sMGAGGGGSAEQLDALVKKDKVVVFLKG
TPEQPQCGFSNAVVQILRLHGVRDYAAY
NVLDDPELRQGIKDYSNWPTIPQVYLNG
EFVGGCDILLQMHQNGDLVEELKKLGIH
SALLDE
|
|
Host:Native protein: E.coli BL21(DE3)-R3, SeMet labelled: E. coli 834(DE3)
|
|
Growth medium and induction protocol:
Native protein: 3 x1 litre TB supplemented with 50 µg/ml Kanamycin and 34 µg/ml Chloramphenicol were inoculated with 10 ml overnight culture and grown at 37°C until OD600 reached 1.5. The temperature was then decreased to 18°C and the protein expression induced with 0.1 mM IPTG over night. The cells were collected by centrifugation and frozen at -80°C.
SeMet labelled: A glycerol stock was used to inoculate 4x50 ml cultures of B834(DE3)/GLRX5A in LB supplemented with 50 µg/ml Kanamycin. The cultures were grown at 25°C until OD600 reached 1.0. The cells were then pelleted, washed 3 times in 100 ml of sterile water and resuspended in 4x10 ml of M9 minimal media (Molecular dimensions Ltd). The cultures were used to innoculate 4x1 litre pre-warmed minimal media supplemented with 40 mg/L SeMet, 50 µg/ml Kanamycin and 100 µM FeCl3 and grown at 37°C until OD600 reached 0.8. The temperature was then decreased to 18°C and the protein expression induced with 0.5 mM IPTG over night. The cells were collected by centrifugation and frozen at -80°C.
|
|
Extraction buffer, extraction method: Lysis buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 20 mM Imidazole, 5% glycerol, Complete® protease inhibitors (1 tablet/50 ml) and 5 U/ml of Benzonase. Cell pellet from 3 respectively 4 litre culture (native and SeMet labelled GLRX5) were resuspended in a total volume of 200 ml lysis buffer. The cells were disrupted by sonication and nucleic acids and cell debris removed by adding 0.15% PEI, followed by centrifugation for 45 minutes at 40000xg. The supernatant was further clarified by filtration (0.45 µm).
|
|
Column 1: Ni-Sepharose FF, 4 ml (GE/Amersham Biosciences)
|
|
Buffers: Binding buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 20 mM Imidazole, 5% glycerol; Wash and elution buffers: 50 mM HEPES pH 7.5, 500 mM NaCl, 5% glycerol, 30 -250 mM imidazole.
|
|
Procedure: The cell lysate was applied onto a 4 ml Ni-Sepharose FF column equilibrated with binding buffer. The column was subsequently washed with 40 ml of binding buffer and the protein eluted using a stepwise gradient of imidazole. All fractions were collected and analysed by SDS-PAGE. GLRX5A had a visible brown color.
|
|
Enzymatic treatment: TEV cleavage.
|
|
Column 3: Ni-Sepharose FF (TEV clean up)
|
|
Buffer: 10 mM HEPES, pH 7.5, 500 mM NaCl, 5% glycerol, 0.5 mM TCEP, 10 mM GSH and 3mM DTT
|
|
Procedure: The protein was concentrated in amicon 3K and the buffer exchanged to 10 mM HEPES, pH 7.5, 500 mM NaCl, 5% glycerol, 0.5 mM TCEP, 10 mM GSH and 3mM DTT. The histidine-tag was then cleaved with 150 µg of TEV protease per 10 mg protein at 4°C for 16 hours. The protein was applied to a 0.4 ml Ni-NTA column and the flow through collected. The column was washed with 5 ml buffer and the flow-through and the wash fraction were analysed by SDS-PAGE.
|
|
Concentration: The protein was concentrated in Amicon (3 K) to 36.6 mg/ml (SeMet) and 93 mg/ml (native). The protein concentration was determined spectrophotometrically using the predicted molar extinction coefficient 11460 (M-1 cm-1).
|
|
UV-Visible Spectroscopy: GLRX5A had an visible brown colour and absorbed at 320 and 420 nm when analysed by UV-Vis spectroscopy.
|
|
Mass spec characterization: The mass of the native and SeMet labelled protein was determined with ESI-MS to 12852.2 and 12946.36 Da, respectively , both in agreement with the predicted mass of the protein.
|
|
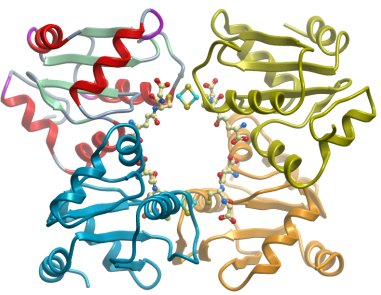
Crystallisation: Phases were obtained from a SeMet substituted crystal grown by vapor diffusion at 20ºC in a sitting drop consisting of 75 nL protein (36.6 mg/ml) and 75 nL well solution containing 70% MPD and 0.1 M Hepes pH 7.5. The structure was refined against native data from a crystal grown by vapor diffusion at 20ºC in a sitting drop of 150 nL protein and 150 nL well solution containing 50% PEG300, 0.2 M MgCl2, 0.1 M cacodylate pH 6.5 and 0.01 M spermine tetrahydrochloride . Both crystals used were of a thin, rod like morphology and visibly brown. The crystals were flash cooled in liquid nitrogen straight from the drop without additional cryo protection.
|
|
Data Collection and Refinement: Both the SeMet and the high resolution data were collected at beam line X10SA at the Swiss light source. The SeMet crystal diffracted to 2.7 Å and was collected at λ=0.9794 Å. After processing in Mosflm the data were prepared in XPREP and 4 SeMet sites were found using SHELXD with an anomalous signal to 3.1 Å. Initial phases were calculated with SHELXE and the space group pinpointed to P43212. These were improved in a manual SHARP run and Buccaneer could be used to build fragments of secondary structural elements into the solvent flattened maps. Positions for four molecules of the closest homolog glutaredoxin C1 from Populus tremula x tremuloides (2E7P) were found by manual molecular replacement using the automatically built fragments as a guide. After a crude rebuild in Coot the model could be used for phasing the higher resolution data with Phaser. The native data were collected at λ=0.979Å in 5 wedges along the thin, rod like crystal that showed highly anisotropic diffraction. 20°C from each segment survived radiation damage and could be merged and scaled to a final resolution of 2.4 Å, although due to the anisotropy of the data the final resolution of the structure could be considered as 2.6 Å.
|