Entry Clone Source: MGC |
Entry Clone Accession: IMAGE:5114635 |
SGC Construct ID: CLK3A-c005 |
GenBank GI number: gi|4502885 |
| Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
TACTTCCAATCCATGCAGAGCAGTAAGCGC
AGCAGCCGGAGTGTGGAAGATGACAAGGAG
GGTCACCTGGTGTGCCGGATCGGCGATTGG
CTCCAAGAGCGATATGAGATTGTGGGGAAC
CTGGGTGAAGGCACCTTTGGCAAGGTGGTG
GAGTGCTTGGACCATGCCAGAGGGAAGTCT
CAGGTTGCCCTGAAGATCATCCGCAACGTG
GGCAAGTACCGGGAGGCTGCCCGGCTAGAA
ATCAACGTGCTCAAAAAAATCAAGGAGAAG
GACAAAGAAAACAAGTTCCTGTGTGTCTTG
ATGTCTGACTGGTTCAACTTCCACGGTCAC
ATGTGCATCGCCTTTGAGCTCCTGGGCAAG
AACACCTTTGAGTTCCTGAAGGAGAATAAC
TTCCAGCCTTACCCCCTACCACATGTCCGG
CACATGGCCTACCAGCTCTGCCACGCCCTT
AGATTTCTGCATGAGAATCAGCTGACCCAT
ACAGACTTGAAACCAGAGAACATCCTGTTT
GTGAATTCTGAGTTTGAAACCCTCTACAAT
GAGCACAAGAGCTGTGAGGAGAAGTCAGTG
AAGAACACCAGCATCCGAGTGGCTGACTTT
GGCAGTGCCACATTTGACCATGAGCACCAC
ACCACCATTGTGGCCACCCGTCACTATCGC
CCGCCTGAGGTGATCCTTGAGCTGGGCTGG
GCACAGCCCTGTGACGTCTGGAGCATTGGC
TGCATTCTCTTTGAGTACTACCGGGGCTTC
ACACTCTTCCAGACCCACGAAAACCGAGAG
CACCTGGTGATGATGGAGAAGATCCTAGGG
CCCATCCCATCACACATGATCCACCGTACC
AGGAAGCAGAAATATTTCTACAAAGGGGGC
CTAGTTTGGGATGAGAACAGCTCTGACGGC
CGGTATGTGAAGGAGAACTGCAAACCTCTG
AAGAGTTACATGCTCCAAGACTCCCTGGAG
CACGTGCAGCTGTTTGACCTGATGAGGAGG
ATGTTAGAATTTGACCCTGCCCAGCGCATC
ACACTGGCCGAGGCCCTGCTGCACCCCTTC
TTTGCTGGCTTGACCCCTGAGGAGCGGTCC
TTCCACACCTAAGACAGTAAAGGTGGATA
|
Final protein sequence (tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfqSMQSSKRSS
RSVEDDKEGHLVCRIGDWLQERYEIVGNLG
EGTFGKVVECLDHARGKSQVALKIIRNVGK
YREAARLEINVLKKIKEKDKENKFLCVLMS
DWFNFHGHMCIAFELLGKNTFEFLKENNFQ
PYPLPHVRHMAYQLCHALRFLHENQLTHTD
LKPENILFVNSEFETLYNEHKSCEEKSVKN
TSIRVADFGSATFDHEHHTTIVATRHYRPP
EVILELGWAQPCDVWSIGCILFEYYRGFTL
FQTHENREHLVMMEKILGPIPSHMIHRTRK
QKYFYKGGLVWDENSSDGRYVKENCKPLKS
YMLQDSLEHVQLFDLMRRMLEFDPAQRITL
AEALLHPFFAGLTPEERSFHT |
Tags and additions: Tag sequence: mhhhhhhssgvdlgtenlyfq*s(m). TEV-cleavable (*) N-terminal hexaHis tag. |
| Host: BL21 (DE3) |
Growth medium, induction protocol: 1 ml from a 10 ml overnight culture in LB, 50 µg/ml kanamycin was used to inoculate 1 litre of LB medium containing 50 µg/ml kanamycin. Cultures were grown at 37°C until they reached an OD600 of 0.3 and then cooled to 18°C. Expression was induced for 4 hours using 1 mM IPTG at an OD600 of 0.6. The cells were collected by centrifugation, transferred to 50 ml tubes, resuspended in 30 ml binding buffer, and frozen. Binding buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 5% Glycerol, 50 mM L-Arg and L-Glu. |
| Extraction buffer, extraction method: The frozen cells were thawed on ice and binding buffer (plus 1 mM PMSF) added to a final volume of 50 ml. Cells were lysed using a high pressure cell disruptor. The lysate was centrifuged at 18,500 RPM for 50 minutes and the supernatant collected for purification. |
Column 1: Ion exchange - Nucleic acid removal. DEAE cellulose (DE52, Whatman), 10 g of resin in 2.5 x 20 cm column. The resin was hydrated in 2.5M NaCl, then washed with 20 ml binding buffer prior to loading the sample. |
Column 1 Buffers: Binding buffer: 50 mM HEPES, 500 mM NaCl, 5% Glycerol, 50 mM L-Arg and L-Glu. |
| Column 1 Procedure: Supernatant was applied at gravity flow, followed by a wash with 50 ml binding buffer. The column flow-through was collected. |
Column 2: Ni-affinity. Ni-NTA (Qiagen), 5 ml of 50% slurry in 1.5 x 10 cm column, washed with binding buffer. |
| Column 2 Buffers: Binding buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 5% Glycerol, 50 mM L-Arg and L-Glu. Wash buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 20 mM Imidazole, 5% glycerol, 50 mM L-Arg and L-Glu. Elution buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 50 to 250 mM Imidazole , 5% Glycerol, 50 mM L-Arg and L-Glu. |
| Column 2 Procedure: The flowthrough from column 1 was loaded by gravity flow on the Ni-NTA column. The column was then washed with 3 x 10 ml wash buffer at gravity flow. The protein was eluted by gravity flow by applying 5-ml portions of elution buffer with increasing concentration of imidazole (50 mM, 100 mM, 150 and 250 mM); fractions were collected until essentially all protein was eluted. After elution DTT was added to a final concentration of 10 mM. |
| Enzymatic treatment : (Dephosphorylation and His tag cleavage) Samples containing CLK3 were pooled and 20 µg GST-lambda phosphatase and 20 µg TEV protease added for overnight incubation at 4°C: protein solution contained 10 mM DTT and 0.05 mM MnCl2. For crystallization of phosphorylated CLK3 the protein was only treated with TEV protease |
Column 3: Size Exclusion Chromatography |
Column 3 Buffers: Fractions containing CLK3 collected from IMAC were concentrated and directly applied to a S75 16/60 HiLoad gel filtration column equilibrated in 50 mM Hepes pH 7.5, 500 mM NaCl, 50 mM L-glutamic acid, 50 mM L-arginine. |
| Column 3 Procedure: AKTA-prime |
Column 4: Anion Exchange Chromatography |
Column 4 Buffers: Fractions containing CLK3 collected from SEC were diluted to a final concentration of 50 mM HEPES pH 7.5, 50 mM NaCl and applied to a MonoQ 5/50 GL equilibrated in 50 mM Hepes pH 7.5, 50 mM NaCl. The potein was eluted using an NaCl gradient. |
| Column 4 Procedure: AKTA-express |
| Mass spec characterization: LC- ESI -MS TOF confirmed the correct mass expected for this construct. |
| Intact Mass: Masses of purified proteins were confirmed by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. Proteins were desalted prior to mass spectrometry by rapid elution off a C3 column with a gradient of 5-95% acetonitrile in water with 0.1% formic acid. |
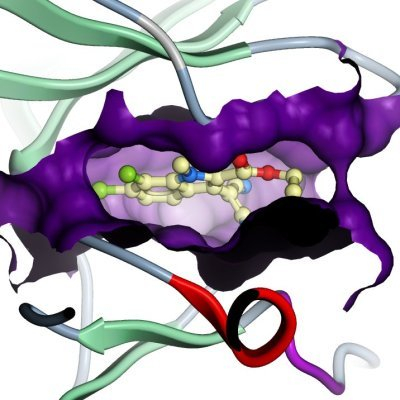
| Crystallization: Crystals were grown at 4°C in 150nl sitting drops mixing 75 nl of CLK3 11.4mg/ml in 50mM Hepes pH 7.5, 200mM NaCl,10mM DTT, with 75 nl of a solution containing 0.2M (NH4)2SO4; 0.1M BIS-TRIS pH 5.5; 25% PEG 3350. The inhibitors were added to the concentrated protein solution (1 mM end concentration) using a DMSO stock solution. |
Data Collection: Resolution: X-ray source: Synchrotron SLS -X10, single wavelength (2wu6) and using Brucker rotating anode (Copper target) generator equipped with a CCD detector (2wu7), respectively. Crystals were cryoprotected using the crystallization solution supplemented with 25% ethylene glycol. |