Entry Clone Source: Synthetic |
Entry Clone Accession: n/a |
SGC Construct ID: GUCY1B3A-c130 |
GenBank GI number: gi|4504215 |
Vector: pNIC-CTHF. Details [ PDF ]; Sequence [ FASTA ] or [ GenBank ]
|
Coding DNA sequence (The DNA sequence was optimized for expression in E. coli and does not match the human gene):
ATGCACAAGCGTCCGGTTCCGGCCAAACG
CTATGACAACGTGACTATTCTGTTCTCCG
GTATCGTAGGGTTTAATGCATTTTGTAGC
AAACATGCGTCGGGCGAGGGTGCTATGAA
GATTGTTAACCTGCTGAATGATCTGTACA
CACGTTTCGATACGCTGACCGACTCTCGC
AAAAACCCGTTTGTGTATAAAGTCGAAAC
TGTTGGCGATAAGTATATGACAGTGAGTG
GTCTGCCGGAGCCGTGCATCCATCACGCC
CGTTCAATTTGTCATCTGGCACTGGACAT
GATGGAAATTGCGGGGCAGGTACAGGTCG
ATGGCGAAAGCGTTCAAATCACGATTGGT
ATCCACACCGGGGAGGTGGTAACTGGCGT
CATTGGTCAGCGCATGCCGCGTTACTGCC
TGTTCGGCAATACGGTTAACCTGACCTCC
CGCACAGAAACCACTGGTGAAAAAGGGAA
AATCAATGTGTCGGAGTATACATACCGTT
GTCTGATGTCTCCGGAAAACAGTGACCCG
CAATTTCATCTGGAGCACCGCGGCCCGGT
AAGCATGAAGGGTAAAAAAGAACCGATGC
AGGTCTGGTTCCTGTCACGTAAGAATACG
GGCACCGAAGAGACTAAACAGGATGATGA
CGCAGAGAACCTCTACTTCCAATCGCACC
ATCATCACCACCATGATTACAAGGATGAC
GACGATAAGTGA |
Tags and additions: C-terminal tag: aenlyfq*shhhhhhdykddddk |
Host cells: BL21(DE3)-R3-Chaperones |
Final protein sequence (tag sequence in lowercase):
MHKRPVPAKRYDNVTILFSGIVGFNAFCS
KHASGEGAMKIVNLLNDLYTRFDTLTDSR
KNPFVYKVETVGDKYMTVSGLPEPCIHHA
RSICHLALDMMEIAGQVQVDGESVQITIG
IHTGEVVTGVIGQRMPRYCLFGNTVNLTS
RTETTGEKGKINVSEYTYRCLMSPENSDP
QFHLEHRGPVSMKGKKEPMQVWFLSRKNT
GTEETKQDDDaenlyfq*shhhhhhdykd
dddk
The asterisk indicates the site of cleavage by TEV protease. This construct includes amino acids 408-619 of GUCY1B3. |
Cell growth and induction: To assist recovery of soluble protein, the expression vector was transformed into BL21(DE3)-R3 cells containing plasmid pGro7 (TAKARA), which provides arabinose-inducible expression of the bacterial chaperone genes GroEL and GroES.
The expression plasmid was transformed into the host strain and plated on LB-agar containing 50 µg/ml kanamycin and 35 µg/ml chloramphenicol. Several colonies were combined to inoculate a 1-ml culture in TB (+ 50 µg/ml kanamycin, 35 µg/ml chloramphenicol). The culture was grown overnight, glycerol was added to 15% v/v (from a 60% stock), and the resulting glycerol stock was frozen at -80°C in 100 µl aliquots. A loop full of cells from the glycerol stock was inoculated into 100-ml of TB medium containing 50 µg/ml kanamycin and 35 µg/ml chloramphenicol and grown overnight at 37°C.
Four cultures of 1-L each of TB medium (+ 50 µg/ml kanamycin) in 2.5L UltraYield baffled flasks were inoculated with 10 ml each of the overnight culture. The cultures were grown at 37°C for 3 hours until OD600 of 1; expression of the chaperone proteins was then induced with 0.2% arabinose for 1 hour. The cultures were then transferred to 18°C, and after one hour IPTG was added to 0.5 mM. Growth was continued overnight. The cells were collected by centrifugation, the pellets were scraped out and transferred to 50-ml Falcon tubes and frozen at -80°C. |
Cell extraction: Lysis buffer: 50 mM HEPES buffer, pH 7.4, 500 mM NaCl, 5 mM imidazole, 5% glycerol, 0.5 mM TCEP, 1x Protease Inhibitors Cocktail Set VII (Calbiochem, 1/1000 dilution), and 15 units/ml Benzonase. 2x Lysis buffer contains the same components at double concentration.
Frozen cell pellets (86 g) were thawed briefly in a bath of warm water (20 - 37°C) then transferred to ice. One volume (i.e. 1 ml for every gram of cells) of 2x lysis buffer was added, followed by 1x lysis buffer to a total volume of 300-ml. The cells were resuspended by agitating and disrupted by sonication. Nucleic acids and cell debris were removed by adding 0.15% PEI (polyethyleneimine) from a 5% (w/v, pH 7.5) stock, stirring for 15 minutes, then centrifugation for 1 hour at 25,000 xg. |
Column 1: Histrap FF 5 ml (GE Healthcare) |
Buffers:
Affinity buffer: 50 mM HEPES buffer, pH 7.4, 500 mM NaCl, 50 mM imidazole, 5% glycerol, 0.5 mM TCEP;
Wash buffer: 50 mM HEPES buffer, pH 7.5, 500 mM NaCl, 25 mM imidazole, 5% glycerol, 0.5 mM TCEP;
Elution buffer: 50 mM HEPES buffer, pH 7.5, 500 mM NaCl, 250 mM imidazole, 5% glycerol, 0.5 mM TCEP. |
Procedure: The cell extract was loaded onto the column at 5 ml/minute on an AKTA-express system (GE Healthcare). The column was then washed with 10 volumes of loading buffer, 10 volumes of wash buffer, then eluted with elution buffer at 4 ml/min. The eluted peak of A280 was automatically collected. |
Column 2: Gel filtration, HiLoad 16/60 Superdex Superdex 75, 120 ml. |
Buffer: GF Buffer: 50 mM HEPES, pH 7.4, 500 mM NaCl, 5% glycerol, 0.5 mM TCEP. |
Procedure: The eluted fraction was loaded and fractionated on the gel filtration column in GF buffer at 1.2 ml/min. 2-ml fractions were collected at the A280 peaks. The fractions were analyzed by SDS-PAGE and relevant fractions were pooled. This column resulted in good separation of the GUCY1B3 protein from the major peak of the chaperon proteins. The elution volume of GUCY1B3 corresponded to approximately 40 kDa, which is compatible with a dimer. |
TEV cleavage and re-purification: GUCY1B3A protein was treated with TEV protease (His6-tagged) overnight at 4°C; the protease and other impurities were removed by passing through NiNTA column. The protein was then concentrated to 12 mg/ml (concentrations estimated by A280, using an extinction coefficient of 17420) |
Protein characterization: ESI-TOF Mass spectrometry revealed a mass of 24,756.2 Da, compared to the predicted 24,755.3 (after tag cleavage). |
Crystallization: Crystals were grown by vapor diffusion at 4°C in 150nl sitting drops. The drops were prepared by mixing 100nl of protein solution and 50nl of precipitant consisting of 0.1M HEPES, pH 7.5, 1.4 M Na3 citrate. The crystals were cryo-protected using the well solution supplemented with 25% ethylene glycol and flash-frozen in liquid nitrogen. |
Data collection: Diffraction data were collected from a single crystal at the Diamond synchrotron beamline I02, at a wavelength of 0.9795 Å. |
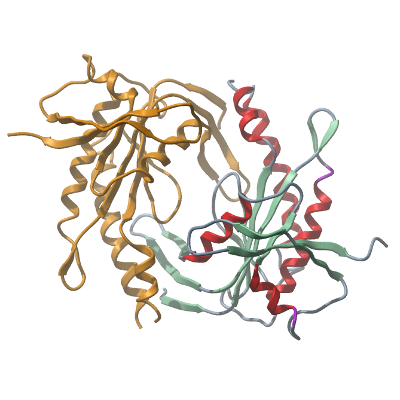
Structure determination: The protein crystallized in space group C2221 with two molecules in the asymmetric unit. The data was phased using PDB: 3ET6 as a model. The model was refined to a final resolution of 1.63 Å, R=18.3, Rfree=21.6. |