Entry Clone Source: Collaborator |
Entry Clone Accession: n/a |
SGC Construct ID: CUL5A-c001 |
GenBank GI number: gi|38257756 |
Vector: pNIC-CTHF. Details [ PDF ]; Sequence [ FASTA ] or [ GenBank ]
|
Coding DNA sequence:
CTGGTTCCGCGTGGATCCAATGTGCAGGG
ACATCTTCCCCTCGTACCACGAGGTTCAA
TGGCGCGCCAGATGGCGACGTCTAATCTG
TTAAAGAATAAAGGTTCTCTCCAGTTTGA
AGACAAGTGGGACTTCATGCATCCAATTG
TTTTGAAGCTTTTACGCCAGGAATCTGTA
ACAAAACAGCAGTGGTTTGATCTATTTTC
GGATGTACATGCTGTCTGTCTCTGGGATG
ATAAAGGCTCATCAAAAATTCATCAGGCT
TTAAAAGAAGATATTCTTGAGTTTATTAA
GCAAGCACAGGCTCGTGTACTGAGCCATC
AAGATGACACAGCTTTGCTGAAGGCATAT
ATTGTTGAATGGCGGAAATTCTTCACACA
GTGTGATATTTTACCAAAACCTTTTTGTC
AATTAGAGGTGACTCTATTGGGTAAACAA
AGCAGCAATAAAAAATCAAATATGGAAGA
CAGTATTGTTCGAAAGCTCATGCTTGATA
CGTGGAATGAGTCGATTTTTTCAAATATA
AAGAACAGACTCCAGGACAGTGCAATGAA
GCTGGTGCATGCTGAGAGATTAGGGGAAG
CTTTTGATTCCCAGCTGGTCATCGGGGTG
CGAGAGTCCTATGTTAATCTTTGCTCCAA
CCCCGAGGACAAGCTTCAGATCTATAGGG
ATAATTTTGAGAAGGCATACTTGGACTCA
ACAGAGAGGTTTTATAGAACACAGGCACC
CTCATATTTACAGCAAAATGGTGTGCAGA
ATTACATGAAATATGCAGATGCTAAATTA
AAAGAAGAAGAAAAAAGAGCACTCCGATA
CTTAGAAACACGACGAGAGTGTAACTCTG
TGGAAGCACTCATGGAATGCTGTGTAAAT
GCGCTGGTGACCTCCTTTAAAGAGACTAT
TTTAGCAGAATGCCAAGGCATGATCAAGC
GAAATGAAACTGAAAAGTTACATTTGATG
TTTTCCTTGATGGACAAAGTTCCTAATGG
GATAGAGCCGATGTTGAAGGACTTGGAGG
AGCATATTATAAGTGCGGGCCTAGCAGAC
ATGGTGGCCGCAGCTGAAACCATCACTAC
TGACTCTGAGAAGTATCGGGAGCAATTAG
ACACACTGTTTAATAGATTCAGTAAACTG
GTCAAAGAAGCTTTTCAGGATGATCCTCG
TTTCCTTACTGCAAGAGATAAGGCATATA
AAGCAGTTGTTAATGATGCTACTATATTT
AAATAAGCGGCCGC |
Tags and additions:
enlyfq*shhhhhhdykddddk, TEV-cleavable (*) C-terminal hexahistidine and FLAG tag. |
Host: BL21(DE3)-R3-pRARE2 (Native) and B834(DE3) (Selenomethionine labelling) |
Expressed protein sequence (tag sequence in lowercase):
MATSNLLKNKGSLQFEDKWDFMHPIVLKL
LRQESVTKQQWFDLFSDVHAVCLWDDKGS
SKIHQALKEDILEFIKQAQARVLSHQDDT
ALLKAYIVEWRKFFTQCDILPKPFCQLEV
TLLGKQSSNKKSNMEDSIVRKLMLDTWNE
SIFSNIKNRLQDSAMKLVHAERLGEAFDS
QLVIGVRESYVNLCSNPEDKLQIYRDNFE
KAYLDSTERFYRTQAPSYLQQNGVQNYMK
YADAKLKEEEKRALRYLETRRECNSVEAL
MECCVNALVTSFKETILAECQGMIKRNET
EKLHLMFSLMDKVPNGIEPMLKDLEEHII
SAGLADMVAAAETITTDSEKYREQLDTLF
NRFSKLVKEAFQDDPRFLTARDKAYKAVV
NDATIFKAenlyfq*shhhhhhdykdddd
k |
Growth medium, induction protocol: A glycerol stock was used to inoculate a 10ml starter culture containing LB media with 50µg/ml Kanamycin and 34 µg/ml chloramphenicol. The starter culture was grown overnight at 37°C with shaking at 200 rpm. The following morning, four flasks containing 1 L LB/kanamycin/chloramphenicol were each inoculated with 5 ml of the starter culture. Cultures were incubated at 37°C with shaking at 180 rpm until an OD 600 ≥ 0.7 was reached. The flasks were then cooled down to 18°C and 0.5mM IPTG added to induce protein expression overnight. Cells were harvested by centrifugation at 4500 rpm at 4°C for 15 min. Cell pellets from each flask were resuspended in 30ml binding buffer (50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole), transferred to 50 ml tubes, and stored at -20°C. The protocol was repeated for subsequent selenomethionine labeling using a 6L expression from the B834(DE3) strain, including 40 mg/L L-selenomethionine. |
Extraction buffer, extraction method: The frozen cells were thawed and 0.5mM TCEP, 1mM PMSF, 50mM L-Arginine, 50 mM L-Glutamic acid added to the cell suspension. The cells were lysed by ultrasonication over 12 min with the sonicator pulsing ON for 5 sec and OFF for 15 sec. The cell lysate was spun down by centrifugation at 17000 rpm at 4°C for 1 h. The supernatant was recovered for purification. |
Column 1: Ni-Affinity Chromatography.
5ml of 50 % Ni-sepharose slurry (Amersham) was applied onto a 1.5 x 10 cm column. The column was first washed with deionised distilled H2O, and then equilibrated with binding buffer. |
Buffers:
Binding buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole;
Wash buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 30mM Imidazole;
Elution buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 50 to 250mM Imidazole. |
Procedure: The supernatant was applied by gravity flow onto the Ni-sepharose column. The bound protein was eluted by applying a step gradient of imidazole (5 ml fractions of elution buffer supplemented with 50mM, 100mM, 150mM and 250mM imidazole). 10mM DTT was added to each fraction collected for overnight storage at 4°C. |
Enzymatic treatment: TEV protease cleavage. Fractions containing CUL5 were treated with TEV protease overnight at 4°C. |
Column 2: Size Exclusion Chromatography – S200 HiLoad 16/60 Superdex run on ÄKTA-Express. |
Buffer: Gel Filtration buffer: 300mM NaCl, 50mM HEPES pH 7.5, 0.5mM TCEP |
Procedure: Prior to applying the protein, the S200 16/60 column was washed and equilibrated with gel filtration buffer. Eluted protein from the Ni-sepharose column was cleaved with TEV protease and pooled and concentrated to 5ml using an Amicon Ultra-15 filter with a 30kDa cut-off. The concentrated protein was directly applied onto the equilibrated S200 16/60 column, and run at a flow-rate of 1 ml/min. The protein was eluted at 90 – 110 ml. Fractions containing the protein were pooled together, and 10mM DTT was added for overnight storage at 4°C. |
Column 3: Ni-Affinity Chromatography.
500µl of 50 % Ni-sepharose slurry was applied onto a Biorad Poly-Prep disposable drip column. The column resin was first washed with deionised distilled H2O, and then equilibrated with binding buffer. |
Buffers:
Binding buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole;
Wash buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 30mM Imidazole;
Elution buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 50 to 250mM Imidazole |
Procedure: The fractions containing CUL5 from gel filtration were applied by gravity flow onto the Ni-sepharose column. The flow through was collected, washed the column with 10ml Wash buffer and the bound impurities were eluted with 500µl of elution buffer with 250mM Imidazole. 10mM DTT was added to the flow through. |
Concentration: The protein was concentrated in an Amicon Ultra-4 filter with a 30 kDa cut-off. |
Mass spectrometry characterization: The purified native protein was homogeneous and had an experimental mass after tag cleavage of 45335.8 consistent with loss of the N-terminal methionine (calculated MW of this species = 45333.8). For the selenomethionine-labelled protein an experimental mass of 45853.8 kDa was observed, as expected for the incorporation of 11 selenomethionine residues. Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. |
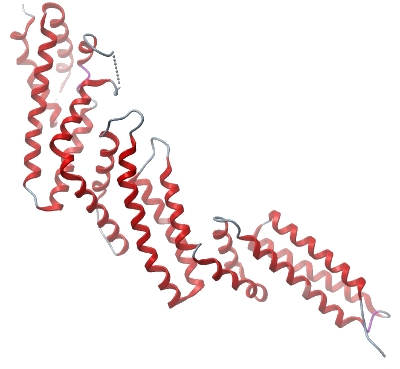
Crystallisation: Protein was buffered in 50mM HEPES, pH 7.5, 300mM NaCl, 10mM DTT, 0.5 mM TCEP, 10 mM L-arginine, 10 mM L-glutamic acid. Protein was concentrated to 16 mg/ml (calculated using extinction co-efficient of 45380). Native crystals were grown at 20°C in 150 nl sitting drops mixing 50 nl protein solution with 100 nl of a reservoir solution containing 0.1M BisTris pH 6.5, 25% PEG 3350, 0.15M NH 4 SO2. On mounting crystals were cryo-protected with an additional 25% ethylene glycol.
Crystals of the selenomethionine-labelled protein (16 mg/ml) were grown at 20°C in 150 nl sitting drops mixing 75 nl of protein solution with 75 nl of reservoir solution containing 20% jeffamine ED-2001 reagent pH 7.0, 0.1 M HEPES pH 6.8. |
Data Collection: Resolution: 2.05Å, X-ray source: Diamond I02 (native) and IO3 (selenomethionine). The native crystal diffracted to a resolution of 2.05 Å and the selenomethionine-derivatized crystals diffracted to 2.27 Å. Both datasets were collected at 100 K. Data were indexed and integrated using iMOSFLM and scaled using SCALA. Phases were found using selenomethionine SAD data and extended to the highest resolution with SOLVE. |