Entry Clone Source: MGC |
Entry Clone Accession: IMAGE:3342973 |
SGC Construct ID: MACROD1A-c010 |
GenBank GI number: gi|13569840 |
Vector: pNH-TrxT. Details [PDF]; Sequence [ FASTA] or [GenBank]
|
Host cells: BL21 (DE3)-R3-pRARE (a phage-resistant derivative of Rosetta2 [Novagen]) |
Coding DNA sequence:
CATATGCACCATCATCATCATCATTCTTCT
GGTATGAGCGATAAAATTATTCACCTGACT
GACGACAGTTTTGACACGGATGTACTCAAA
GCGGACGGGGCGATCCTCGTCGATTTCTGG
GCAGAGTGGTGCGGTCCGTGCAAAATGATC
GCCCCGATTCTGGATGAAATCGCTGACGAA
TATCAGGGCAAACTGACCGTTGCAAAACTG
AACATCGATCAAAACCCTGGCACTGCGCCG
AAATATGGCATCCGTGGTATCCCGACTCTG
CTGCTGTTCAAAAACGGTGAAGTGGCGGCA
ACCAAAGTGGGCGCACTGTCTAAAGGTCAG
TTGAAAGAGTTCCTCGACGCTAACCTGGCC
GGTACCGAGAACTTGTACTTCCAATCCATG
TCGGCGGGAGTTGGGGCGTGGGGGGCGGCG
GCGGTGGGGCGGACAGCCGGGGTGCGCACT
TGGGCCCCCCTGGCCATGGCGGCGAAGGTG
GACCTGAGCACCTCCACCGACTGGAAGGAG
GCGAAATCCTTTCTGAAGGGCCTGAGTGAC
AAGCAGCGGGAGGAACATTACTTCTGCAAG
GACTTTGTCAGGCTGAAGAAGATCCCGACA
TGGAAGGAGATGGCGAAAGGGGTGGCTGTG
AAGGTGGAGGAGCCCAGGTATAAAAAGGAC
AAGCAGCTCAATGAGAAAATCTCCCTGCTC
CGCAGCGACATCACCAAGCTGGAGGTGGAC
GCCATCGTCAACGCCGCCAACAGCTCCCTG
CTCGGAGGCGGTGGCGTGGACGGCTGCATT
CATCGGGCCGCCGGCCCCCTGCTTACCGAC
GAGTGCCGGACCCTGCAGAGCTGTAAGACT
GGCAAGGCCAAGATCACCGGCGGCTATCGG
CTCCCGGCCAAGTACGTCATCCACACAGTG
GGGCCCATCGCCTACGGGGAGCCCAGCGCC
AGCCAGGCTGCCGAGCTCCGCAGCTGCTAC
CTGAGCAGTCTGGACCTGCTGCTGGAGCAC
CGGCTCCGCTCGGTGGCGTTCCCCTGCATC
TCCACCGGCGTGTTTGGCTACCCCTGTGAG
GCGGCCGCCGAGATCGTGCTGGCCACGCTG
CGAGAGTGGCTGGAGCAGCACAAGGACAAG
GTGGACCGGCTGATCATCTGCGTGTTCCTC
GAGAAGGACGAGGACATCTACCGGAGCCGG
CTCCCCCACTACTTCCCCGTGGCCTGACAG
TAAAGGTGGATACGGATCCGAATTCGAGCT
CCGTCGACAAGCTT
|
Tags and additions: N-terminal, TEV cleavable His6 and thioredoxin tag.
MHHHHHHSSGMSDKIIHLTDDSFDTDVLKA
DGAILVDFWAEWCGPCKMIAPILDEIADEY
QGKLTVAKLNIDQNPGTAPKYGIRGIPTLL
LFKNGEVAATKVGALSKGQLKEFLDANLAG
TENLYFQ*SM |
Protein sequence without tag:
SMSAGVGAWGAAAVGRTAGVRTWAPLAMAA
KVDLSTSTDWKEAKSFLKGLSDKQREEHYF
CKDFVRLKKIPTWKEMAKGVAVKVEEPRYK
KDKQLNEKISLLRSDITKLEVDAIVNAANS
SLLGGGGVDGCIHRAAGPLLTDECRTLQSC
KTGKAKITGGYRLPAKYVIHTVGPIAYGEP
SASQAAELRSCYLSSLDLLLEHRLRSVAFP
CISTGVFGYPCEAAAEIVLATLREWLEQHK
DKVDRLIICVFLEKDEDIYRSRLPHYFPVA |
Final protein sequence:
AMAAKVDLSTSTDWKEAKSFLKGLSDKQRE
EHYFCKDFVRLKKIPTWKEMAKGVAVKVEE
PRYKKDKQLNEKISLLRSDITKLEVDAIVN
AANSSLLGGGGVDGCIHRAAGPLLTDECRT
LQSCKTGKAKITGGYRLPAKYVIHTVGPIA
YGEPSASQAAELRSCYLSSLDLLLEHRLRS
VAFPCISTGVFGYPCEAAAEIVLATLREWL
EQHKDKVDRLIICVFLEKDEDIYRSRLPHY
FPVA
The expression construct included aa 58-325 of MACROD1 downstream of the fusion tag. The mass of the protein following cleavage of the tag by TEV protease was lower than expected; mass spectometry indicated that the protein was lacking 25 aa downstream of the TEV protease cleavage site, resulting in a protein encompassing aa 82-325 of MACROD1
|
Cell growth and induction:
The expression plasmid was transformed into the host strain and plated on LB-agar containing 50 µg/ml kanamycin and 35 µg/ml chloramphenicol. Several colonies were combined to inoculate a 1-ml culture in
LB (+ 50 µg/ml kanamycin, 35 µg/ml chloramphenicol). The culture was grown overnight, glycerol was added to 15% v/v (from a 60% stock), and the resulting glycerol stock was frozen at -80°C in 100 µl aliquots. A
loop full of cells from the glycerol stock was inoculated into 60-ml of LB medium containing 50 µg/ml kanamycin and 35 µg/ml chloramphenicol and grown overnight at 37°C.
Four cultures of 1-L each of TB medium (+ 50 µg/ml kanamycin) in 2.5L UltraYield baffled flasks were inoculated with 15 ml each of the overnight culture. The cultures were grown at 37°C for 4 hours until
OD600 of 1. The cultures were then transferred to 19°C, and after 45 minutes IPTG was added to 0.75 mM. Growth was continued overnight. The cells were collected by centrifugation, the pellets were resuspended in 105 ml of Lysis
Buffer with the addition of a 1:5000 dilution of Calbiochem Protease inhibitor set VII. The resuspended cell pellet was placed in a -80°C freezer.
Lysis Buffer: 50 mM Hepes pH 7.4, 500 mM NaCl, 5% Glycerol, 5 mM Imidazole pH 7.4, 0.5 mM TCEP |
Cell Lysis: The re-suspended cell pellet was lysed by sonication. PEI (polyethyleneimine) was added to a final concentration of 0.15 % from a 5% (w/v, pH 7.5) stock, stirring for 15
minutes, then centrifugation for 45 minutes at 25,000 xg. |
Column 1: Histrap FF 5 ml (GE Healthcare) |
Column 1 Buffers:
Binding Buffer: 50 mM HEPES pH 7.4, 500 mM NaCl, 5% Glycerol, 5 mM Imidazole pH 7.4, 1.0 mM TCEP
Wash Buffer: 50 mM HEPES pH 7.4, 500 mM NaCl, 5% Glycerol, 30 mM Imidazole pH 7.4, 1.0mM TCEP
Elution Buffer: 50 mM HEPES pH 7.4, 500 mM NaCl, 5% Glycerol, 300 mM Imidazole pH 7.4, 1.0 mM TCEP |
Column 1 Procedure: The cell extract was loaded onto the column at 5 ml/minute on an AKTA-express system (GE Healthcare). The column was then washed with 10 volumes of binding
buffer, 10 volumes of wash buffer, then eluted with elution buffer at 4 ml/min. The eluted peak of A280 was automatically collected. |
Column 2: Gel Filtration, HiLoad 16/60 SUperdex 75 (120 ml) |
Column 2 Buffer: GF Buffer: 20 mM HEPES pH 7.5, 250 mM NaCl, 5% glycerol; 1.0 mM TCEP |
Column 2 Procedure: The eluted fraction was loaded and fractionated on the gel filtration column in GF buffer at 1.2 ml/min. 1.75-ml fractions were collected at the
A280 peaks. The fractions were analyzed by SDS-PAGE and relevant fractions were further analyzed by Mass spectrometry (ESI-TOF). |
Tag cleavage: The HIS- TrxT tag was designed to be cleavable by Tobacco Etch Virus (TEV) and fractions which by gel indicated full length protein and a mixture of full length and
shorter protein were pooled and treated by adding TEV protease (7.4mg/ml) at a mass ratio of 1:30 to MACROD1. Removal of the HIS/TrxT tag, the TEV protease and any other contaminating proteins was achieved through batch binding with
5ml bed volume of Ni-NTA. |
Column 3: 5ml Ni-NTA batch bind |
Column 3 Procedure: The pooled fractions (containing the full length and mixed smaller already auto cleaved fractions) were batch bound to 5ml of Ni-NTA pre-equilibrated with GF
buffer, and the flow through collected. |
Column 4: Gel Filtration, HiLoad 16/60 SUperdex 75 (120 ml) |
Column 4 Buffers: GF Buffer: 20 mM HEPES pH 7.5, 250 mM NaCl, 5% glycerol; 1.0 mM TCEP |
Column 4 Procedure: The flow through from column 3 was collected and concentrated to 3ml using a 10kDa MW CO spin concentrator and injected in a 5ml loop4 on the AKTA Xpress. The
concentrated sample was automatically injected onto an S75 16/60 column pre-equilibrated in GF Buffer at 1.2 ml/min. 1.75 ml fractions were collected and analyzed by SDS-PAGE. The fractions containing MACROD1 were pooled and
concentrated to 76mg/ml using a 10kDa MW CO spin concentrator, then frozen in liquid N2 and stored at -80°C. |
Mass spectrometry characterization: Mass analysis by electrospray-ionization time-of0flight (ESI-TOF) indicated an intact mass of 27206.0357, which is lower than the predicted mass
of 29688.4 Da. MS/MS analysis of peptides following tryptic digest identified the protein as MACROD1. It appears that the protein was cleaved during purification, leading to loss of a further 26 amino acids downstream of the TEV
protease cleavage site. |
Dilution of protein prior to crystallisation: The protein was found to be too concentrated using a PCT test and diluted to 20mg/ml working concentration using GF buffer (20 mM HEPES
pH 7.5, 250 mM NaCl, 5% glycerol; 1.0 mM TCEP).
|
Crystallisation: Crystals grew from a ratio of 1:2, 1:1 and 2:1 protein to precipitant solution (0.2M (NH4)2SO4; 0.1M HEPES pH 7.5; 25% PEG 3350
4°C), using the vapour diffusion method.
|
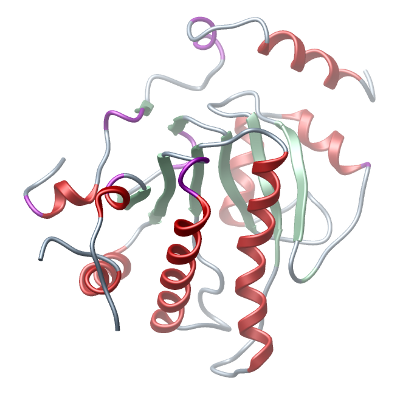
Structure determination: A crystal was cryo protected with a mixture of 1 ul reservoir solution with 1 ul 100% PEG 300 which was added to the drop containing the crystal. This
corresponds to a PEG 300 concentration of 50%. The crystal was flash frozen in liquid nitrogen and all measurements were carried out at 100K. Data collection was performed at Diamond beamline IO3 at a wavelength of 0.9795 Å. The
structure with the PDB ID 1SPV was used as a model for phasing in molecular replacement The structure was refined to 1.7 Å resolution and Rwork/Rfree (%) of 16.4/20.8. |