Entry Clone Source: Synthetic |
Entry Clone Accession: n/a |
SGC Construct ID: LOC340156A-c049 |
GenBank GI number: gi|42657507 |
| Vector: pFB-LIC-Bse. Details [ PDF ]; Sequence [ FASTA ] or [ GenBank ]
|
Coding DNA sequence:
TACTTCCAATCCATGAAGAACTCCGGTGAT
CAGGCCATGGGCCACCATCATCATCATCAT
TCTTCTGGTGTAGATCTGGGTACCGAGAAC
CTGTACTTCCAATCCATGAAGAACTCCGGT
GATCAGGACTCTCGTTCAGGCCATAATGAG
GCAAAAGAAGTATGGAGTAATGCGGATTTG
ACTGAGCGCATGCCAGTGAAATCGAAGCGT
ACAAGCGCATTGGCCGTTGACATCCCTGCT
CCGCCCGCGCCATTCGATCACCGCATTGTC
ACCGCAAAACAAGGTGCCGTAAACTCCTTT
TATACTGTGTCTAAAACGGAGATCCTGGGC
GGCGGCCGTTTCGGTCAGGTTCATAAGTGC
GAGGAAACCGCTACTGGCCTGAAATTGGCA
GCCAAGATTATCAAAACACGCGGTATGAAA
GACAAAGAGGAAGTCAAGAACGAGATTTCA
GTGATGAATCAACTGGATCACGCAAACTTG
ATCCAGCTGTACGATGCATTTGAAAGTAAG
AATGATATTGTATTGGTGATGGAATATGTT
GACGGCGGTGAGCTGTTTGATCGTATCATT
GACGAATCGTACAACCTGACCGAGTTGGAT
ACTATCCTGTTCATGAAACAAATCTGTGAA
GGCATTCGCCACATGCACCAAATGTATATC
TTGCATCTGGACCTGAAACCTGAGAACATT
TTGTGCGTCAATCGTGATGCCAAGCAGATC
AAAATTATCGACTTTGGCCTGGCGCGCCGT
TACAAGCCGCGCGAGAAATTGAAAGTAAAC
TTCGGCACCCCCGAGTTTCTGGCACCAGAA
GTGGTTAATTATGATTTCGTCAGCTTCCCT
ACGGATATGTGGTCCGTGGGTGTAATTGCA
TATATGTTGCTGTCTGGCCTGTCACCGTTC
TTGGGTGATAACGACGCCGAAACCCTGAAC
AATATCTTGGCGTGTCGTTGGGACCTTGAG
GACGAGGAGTTCCAAGATATCAGTGAGGAG
GCAAAAGAGTTCATTTCGAAACTGTTGATC
AAGGAGAAAAGCTGGCGCATTTCCGCCTCT
GAAGCACTGAAACACCCCTGGTTGTCAGAC
CATAAGCTGCACAGTCGTTTGTCGGCGCAG
AAAAAAAAGAACCGCGGCAGCGATGCTCAA
GACTTCGTTACAAAATGACAGTAAAGGTGG
ATACGGATCCGAATTCGAGCTCCGTCGACA
AGCTTGCTCAAGACTTCGTTACAAAATGAC
|
Final protein sequence:
MGHHHHHHSSGVDLGTENLYFQ^SMKNSGD
QDSRSGHNEAKEVWSNADLTERMPVKSKRT
SALAVDIPAPPAPFDHRIVTAKQGAVNSFY
TVSKTEILGGGRFGQVHKCEETATGLKLAA
KIIKTRGMKDKEEVKNEISVMNQLDHANLI
QLYDAFESKNDIVLVMEYVDGGELFDRIID
ESYNLTELDTILFMKQICEGIRHMHQMYIL
HLDLKPENILCVNRDAKQIKIIDFGLARRY
KPREKLKVNFGTPEFLAPEVVNYDFVSFPT
DMWSVGVIAYMLLSGLSPFLGDNDAETLNN
ILACRWDLEDEEFQDISEEAKEFISKLLIK
EKSWRISASEALKHPWLSDHKLHSRLSAQK
KKNRGSDAQDFVTK
^ TEV cleave site |
Tags and additions: Cleavable N-terminal His6 tag. |
| Host: SF9 insect cells |
Growth medium, induction protocol:
LOC340156 was expressed in Sf-9 cells using P-III BV stock. The cells were seeded in Sf-9 cells at the density of 2 x 106 cells/ml. Each 2 L polycarbonate flask contained about 750 ml of the culture in Insect-Xpress medium.
The cells in each flask were infected with 7.5 ml of BV stock and incubated at 27°C for 48 hrs. After 48 hrs, cells were harvested by centrifugation at 1800 rpm for 20 min. The cell pellets were washed once with PBS and stored at -80°C for purification at later date. |
Extraction buffer, extraction method:
The cell pellet from 4 L culture was removed from -80°C, thawed rapidly at 37°C and transferred on ice.
The cells were re-suspended in 3 volumes of lysis buffer (50 mM HEPES, 300 mM NaCl, 5% glycerol, pH 7.5, 1 mM TCEP, 1:1000 dilution of protease inhibitor cocktail).
The cell suspension was then sonicated (40% amplitude, pulse of 10 sec on and off for 10 sec) for 5 min and centrifuged at 16,000 rpm for 45 min. |
Column 1: Ni-affinity. Ni-Sepharose (Amersham), 5 ml of 50 % slurry in 1.5 x 10 cm column, washed with binding buffer. |
Procedure: The supernatant was mixed with 10 ml of 50% Ni-NTA slurry. The mixture was rotated for 1 hour at 4°C followed by loading on a column, washing with 150 ml of wash buffer-1 (50 mM HEPES, 300 mM NaCl, 5% glycerol, pH 7.5), washing with 100 ml of wash buffer-2 (50 mM HEPES, 300 mM NaCl, 5% glycerol, pH 7.5, 25 mM imidazole).
The protein was eluted 30 ml of 300 mM imidazole in buffer containing 50 mM HEPES, 300 mM NaCl, 5% glycerol, pH 7.5. Fractions of the eluates were separated on SDS-PAGE and stained with Coomassie blue. |
Column 2: Hi Q-TRAP column |
Buffers:
Buffer A: 50 mM HEPES, 150 mM NaCl, 5% glycerol, pH 7.5
Buffer B: 50 mM HEPES, 2 M NaCl, 5% glycerol, pH 7.5 |
Procedure:
The 30 ml eluate from Ni-NTA was diluted in 50 mM HEPES, 5% glycerol to achieve final NaCl concentration of 150 mM. The resultant 60 ml protein sample was used for anion exchange chromatography using 5 ml HiTrap Q FF column. |
Column 3: Superdex 75 GF column
|
Procedure: The fractions from anion exchange were pooled together and concentrated to 1ml using 10 spin concentrator with a 10 kDa cut off. The concentrated samples were applied onto a Superdex 75 GF column equilibrated in GF Buffer (50 mM HEPES, 300 mM NaCl, 5% glycerol, pH 7.5). |
Mass spectrometry characterization: LC- ESI -MS TOF revealed the expected mass of the protein (42433) as predicted from the sequence of this protein. |
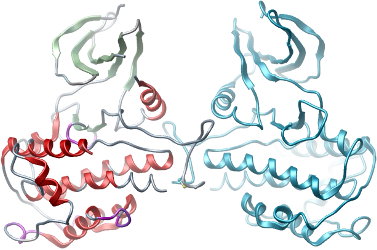
Crystallization: Crystals were grown at 4°C in 300 nl sitting drops from a 2:1 ratio of protein to reservoir solution containing 2M Ammonium Sulfate; 2.5w/v PEG400; in Hepes buffer (50 mM) pH 8.0.
|
Protein concentration: Protein was concentrated to 10 mg/ml using an Amicon 10 kDa cut-off concentrator. |
Data Collection: Crystals were cryo-protected using the well solution supplemented with 2M Li2SO4 and flash frozen in liquid nitrogen.
X-ray source: Diffraction data were collected from a single crystal on Diamond beamline IO2 at a single wavelength of 0.9802 Å and the structure was refined to 2.8 Å.
Phasing: The structure was solved by molecular replacement using the structure of PDB 1KOB as a starting model.
|