Entry Clone Source: Synthetic |
Entry Clone Accession: n/a |
SGC Construct ID: SRPK2A-c003 |
GenBank GI number: gi|33188449 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ]
|
Amplified construct sequence:
TACTTCCAATCCATGGGTAGCGATGACGAG
GAGCAGGAAGATCCCGCCGATTACTGTAAA
GGCGGTTATCATCCAGTGAAGATTGGCGAC
TTGTTCAACGGTCGTTACCACGTTATCCGC
AAACTGGGCTGGGGTCACTTTTCCACTGTC
TGGTTGTGCTGGGACATGCAAGGCAAGCGT
TTCGTAGCTATGAAAGTGGTAAAATCTGCG
CAGCACTATACAGAAACCGCACTGGATGAA
ATTAAGTTGCTGAAATGTGTCCGCGAGTCA
GATCCCTCGGACCCGAATAAGGACATGGTA
GTGCAACTGATCGATGACTTTAAGATTTCG
GGTATGAACGGCATCCATGTTTGCATGGTA
TTCGAAGTGTTGGGTCACCATCTGTTGAAA
TGGATTATCAAGAGCAATTACCAGGGCCTG
CCCGTACGTTGCGTGAAATCCATTATCCGC
CAAGTTTTGCAGGGCCTGGATTATCTGCAC
TCTAAGTGCAAAATCATTCATACTGACATC
AAACCAGAGAACATTTTGATGTGCGTCGAT
GACGCCTACGTACGTCGCATGGCTGCGGAA
GCAACAGAGTGGCAGAAGGCCGGCGCTCCT
CCGCCCTCAGGTAGTGCGGTGTCGACCGCC
CCAGCAGCTGATCTGTTGGTTAATCCTCTG
GACCCGCGTAACGCCGACAAAATCCGCGTC
AAGATTGCGGACCTGGGCAACGCATGCTGG
GTGCACAAACACTTTACTGAAGATATCCAA
ACACGTCAGTATCGCAGCATTGAGGTATTG
ATCGGTGCTGGCTACTCCACCCCAGCCGAC
ATCTGGTCTACTGCGTGTATGGCTTTCGAA
CTGGCAACCGGTGATTATTTGTTCGAACCC
CACTCAGGCGAGGACTACAGTCGTGATGAG
GACCATATTGCGCACATCATTGAGCTGTTG
GGTTCGATCCCTCGCCATTTTGCGCTGAGC
GGCAAATACTCCCGTGAGTTCTTTAATCGC
CGTGGTGAGCTGCGCCACATTACAAAGTTG
AAACCGTGGTCGCTGTTCGATGTTTTGGTC
GAAAAGTATGGCTGGCCACATGAGGACGCT
GCCCAGTTTACCGATTTCCTGATCCCCATG
CTGGAAATGGTGCCTGAGAAACGTGCATCA
GCCGGTGAATGCTTGCGTCACCCGTGGCTG
AACAGTTGACAGTAAAGGTGGATA |
Tags and additions: Cleavable N-terminal His6 tag. |
Final protein sequence (tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfqs^MGSDDEE
QEDPADYCKGGYHPVKIGDLFNGRYHVIRK
LGWGHFSTVWLCWDMQGKRFVAMKVVKSAQ
HYTETALDEIKLLKCVRESDPSDPNKDMVV
QLIDDFKISGMNGIHVCMVFEVLGHHLLKW
IIKSNYQGLPVRCVKSIIRQVLQGLDYLHS
KCKIIHTDIKPENILMCVDDAYVRRMAAEA
TEWQKAGAPPPSGSAVSTAPAADLLVNPLD
PRNADKIRVKIADLGNACWVHKHFTEDIQT
RQYRSIEVLIGAGYSTPADIWSTACMAFEL
ATGDYLFEPHSGEDYSRDEDHIAHIIELLG
SIPRHFALSGKYSREFFNRRGELRHITKLK
PWSLFDVLVEKYGWPHEDAAQFTDFLIPML
EMVPEKRASAGECLRHPWLNS
^ TEV cleave site
|
Host: BL21 (DE3)R3-pRARE2 (Phage resistant strain) |
Growth medium, induction protocol: The expression plasmid was transformed into the host strain and plated on LB-agar containing 50 µg/ml kanamycin and 35 µg/ml chloramphenicol. Several colonies were combined to inoculate a 1-ml culture in TB (+ 50 µg/ml kanamycin, 35 µg/ml chloramphenicol). The culture was grown overnight, glycerol was added to 15% v/v (from a 60% stock), and the resulting glycerol stock was frozen at -80°C in 100 µl aliquots. A loopful of cells from the glycerol stock was inoculated into 5-ml of LB medium containing 100 µg/ml kanamycin and 35 µg/ml chloramphenicol and grown overnight at 37°C. Overnight culture was used to inoculate 1 litre of LB containing 50 µg/ml kanamycin & 34 µg/ml chloramphenicol. Culture was grown at 37 °C until the OD600 reached ~3 and then cooled to 18°C for 1 hour. IPTG was added to 0.1 mM, and growth continued at 18°C overnight. The cells were collected by centrifugation then pellet re-suspended in 2x lysis buffer and frozen. Lysis buffer: 50 mM HEPES buffer, pH 7.5, 500 mM NaCl, 10 mM imidazole, 0.5 mM TCEP, 1x Protease Inhibitors Cocktail Set VII (Calbiochem, 1/1000 dilution), and 15 units/ml Benzonase. 2x Lysis buffer contains the same components at double concentration. |
Extraction buffer, extraction method: Frozen pellets were thawed and fresh 0.5 mM TCEP added to the lysate. Cells were lysed by high pressure homogenization (20 kpsi). The lysate was centrifuged at 16,500 rpm for 60 minutes and the supernatant collected for purification. |
Column 1: Ni-affinity, HisTrap Crude FF, 5 ml (GE Healthcare). |
Column 1 Buffers:
Affinity buffer: 50 mM HEPES buffer, pH 7.5, 500 mM NaCl, 10 mM imidazole, 5% Glycerol, 0.5 mM TCEP.
Wash buffer: 50 mM HEPES buffer, pH 7.5, 500 mM NaCl, 30 mM imidazole, 5% Glycerol, 0.5 mM TCEP
Elution buffer: 50 mM HEPES buffer, pH 7.5, 500 mM NaCl, 300 mM imidazole, 5% Glycerol, 0.5 mM TCEP. |
Column 1 Procedure: The cell extract was loaded on the column at 4 ml/minute on an ÄKTA-express system (GE Healthcare). The column was washed with 10 volumes of lysis buffer, 10 volumes of wash buffer, and then eluted with elution buffer at 4 ml/min. The eluted peak of A280 was automatically collected. |
Column 2: Gel filtration, Hiload 16/60 Superdex S75 prep grade, 120 ml (GE Healthcare) |
Column 2 Buffer: 50 mM HEPES, pH 7.5, 500 mM NaCl, 5% Glycerol, 0.5 mM TCEP. |
Column 2 Procedure: The eluted fractions from the Ni-affinity Histrap column were loaded on the gel filtration column in GF buffer at 1.2 ml/min. Eluted proteins were collected in 2-ml fractions and analyzed on SDS-PAGE. |
TEV protease digestion: Peak fractions containing SRPK2 were pooled and TEV protease was added at a molar ratio of 1:30. The digestion was left overnight at 4 °C. His-TEV and contaminating proteins were removed by passing to 300µl Ni resin pre-equilibrated in GF buffer. The flow through containing TEV-cleaved protein was collected, concentrated to 30 mg/ml using a centricon centrifugal device with a 30kDa MWCO, frozen at -80°C in 500µl aliquots. |
Mass spectrometry characterization: Measured: 44237.55; Expected: 44238 |
Protein Concentration: 4.5 mg of SRPK2 was diluted into 15 ml of low salt buffer (5mM HEPES. pH 7.5, 150 mM NaCl, 1% glycerol and 20 mM DTT). 2 molar excess of ligand was added to diluted SRPK2 and protein-ligand complex was concentrated to 30 mg/ml using an Amicon 30 kDa cut-off concentrator. |
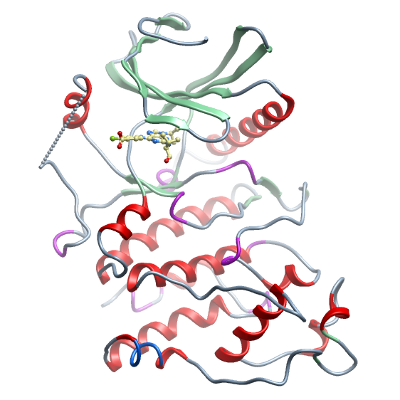
Crystallization: Crystals in complex with the ATP competitive inhibitor Purvalanol B (Sigma catalogue number "P5234") were grown at 4°C in 150nl sitting drops from a 1:1 ratio of reservoir solution (0.5M ammonium sulphate, 0.1M sodium acetate pH4.75) and protein (23mg/ml). Thin rod-shaped crystals appeared after 7-14days. |
Data Collection: Resolution: Crystals were cryo-protected using the well solution supplemented with 35 % ethylene glycol and flash frozen in liquid nitrogen.
X-ray source: Diffraction data were collected from a single crystal to a resolution of 2.5Å on the I24 microfocus beamline at the DIAMOND Light Source. |