The growth hormone releasing hormone receptor, GHRHR, belongs to the class B (Secretin receptor) subfamily of seven transmembrane G-protein-coupled receptor (GPCR) superfamily. It is predominantly expressed in the anterior pituitary gland but also found in hypothalamus, kidney and placenta. GHRHR is essential for normal somatotroph proliferation and for the synthesis and secretion of growth hormone (GH) (Martari and Salvatori, 2009).
Several mutations have been identified to date in the GHRHR gene including nonsense mutations, splice site mutations, microdeletions as well as several different missense mutations and mutations in the promoter region of GHRHR (Castro-Feijoo et al., 2005; Dattani, 2005; Martari and Salvatori, 2009). A naturally occurring mouse model termed the "little mouse" has been found to have a point mutation of Asp60 to Gly in GHRHR, that prevents ligand binding. Mice homozygous for the mutation exhibit reduced growth from 2 weeks of age, impaired growth hormone synthesis and release, obesity, decreased bone mass, reduced fertility in males, impaired first lactations and extended life span (Baumann, 1999; Gertner et al., 1998; Liang et al., 2003). Mutations of the GHRH receptor have been associated with human isolated growth hormone deficiency (IGHD), also known as Dwarfism of Sindh or pituitary dwarfism I, an autosomal recessive disorder characterized by short stature. Another splice variant, which is expressed in normal tissue, is also abundantly expressed in tumors and shows high intrinsic, ligand independent activity (Canzian et al., 2005; Mulhall et al., 2005; Wagner et al., 2006).
Over-expression of GHRHR has been found in some cancers, including triple negative breast cancer, osteosarcomas and Ewing's sarcomas where growth hormone-releasing hormone (GHRH) functions as an autocrine/paracrine growth factor. GHRHR antagonists have been shown to inhibit proliferation of cancer cells. In addition, GHRHR agonists have cardioprotective effects following heart attack in rats, limiting damage to the heart and promoting cell repair, by a mechanism that was independent of growth hormone and IGF mechanisms (Kanashiro-Takeuchi et al., 2010). If this effect is reproduced in humans, then GHRHR agonists have the potential to provide improved treatment for heart attacks.
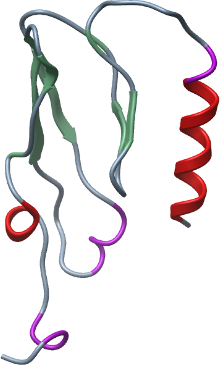
Here we report the structure of the extracellular domain of GHRHR at 1.95 Å resolution.