Entry Clone Source: MGC |
Entry Clone Accession: BC014928 |
SGC Construct ID: MINAB-c001 |
GenBank GI number: gi|23346418 |
Vector: pNIC28-Bsa4. Details [ PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
CATATGCACCATCATCATCATCATTCTTCT
GGTGTAGATCTGGGTACCGAGAACCTGTAC
TTCCAATCCATGGCAGCTGGGGGGCCTTCA
GCTTTAAACTTTGACAGTCCCAGTAGTCTC
TTTGAAAGTTTAATCTCGCCCATCAAGACA
GAGACTTTTTTCAAGGAATTCTGGGAGCAG
AAGCCCCTTCTCATTCAGAGAGATGACCCT
GCACTGGCCACATACTATGGGTCCCTGTTC
AAGCTAACAGATCTGAAGAGTCTGTGCAGC
CGGGGGATGTACTATGGAAGAGATGTGAAT
GTCTGCCGGTGTGTCAATGGGAAGAAGAAG
GTTTTAAATAAAGATGGCAAAGCACACTTT
CTTCAGCTGAGAAAAGATTTTGATCAGAAA
AGGGCAACGATTCAGTTTCACCAACCTCAG
AGATTTAAGGATGAGCTTTGGAGGATCCAG
GAGAAGCTGGAATGTTACTTTGGCTCCTTG
GTTGGCTCGAATGTGTACATAACTCCCGCA
GGATCTCAGGGCCTGCCGCCCCATTATGAT
GATGTCGAGGTTTTCATCCTGCAGCTGGAG
GGAGAGAAACACTGGCGCCTCTACCACCCC
ACTGTGCCCCTGGCACGAGAGTACAGCGTG
GAGGCCGAGGAAAGGATCGGCAGGCCGGTG
CATGAGTTTATGCTGAAGCCGGGTGATTTG
TTGTACTTTCCCAGAGGAACCATTCATCAA
GCGGACACTCCTGCGGGGCTGGCCCACTCG
ACTCACGTGACCATCAGCACCTACCAGAAC
AATTCATGGGGAGATTTCCTTTTGGATACC
ATCTCGGGGCTTGTATTTGATACTGCAAAG
GAAGACGTGGAGTTACGGACCGGCATACCC
CGGCAGCTGCTCCTGCAGGTGGAATCCACA
ACTGTTGCTACAAGACGATTAAGTGGCTTC
CTGAGGACACTTGCAGACCGGCTGGAGGGC
ACCAAAGAACTGCTTTCCTCAGACATGAAG
AAGGATTTTATTATGCACAGACTCCCCCCT
TACTCTGCGGGAGATGGGGCAGAGCTGTCA
ACACCAGGTGGAAAGTTACCGAGGCTGGAC
AGTGTAGTGAGACTGCAGTTTAAAGACCAC
ATTGTCCTCACAGTACTGCCGGATCAAGAT
CAATCTGATGAAACTCAAGAAAAGATGGTG
TACATCTATCATTCCTTAAAGAATAGTAGA
GAGACACACATGATGGGAAATGAGGAGGAA
ACAGAGTTTCATGGACTTCGCTTCCCTTTG
TCACATTTGGATGCACTGAAGCAAATTTGG
AATAGTCCAGCTATTTCTGTCAAGGACCTG
AAACTTACTACAGATGAGGAAAAGGAAAGC
CTGGTATTATCCCTCTGGACAGAATGTTTA
ATTCAAGTAGTCTAGCAGTAAAGGTGGATA
CGGATCCGAA |
Final protein sequence (tag sequence in lowercase)
mhhhhhhssgvdlgtenlyfqsMPKKAKPT
GSGKEEGPAPCKQMKLEAAGGPSALNFDSP
SSLFESLISPIKTETFFKEFWEQKPLLIQR
DDPALATYYGSLFKLTDLKSLCSRGMYYGR
DVNVCRCVNGKKKVLNKDGKAHFLQLRKDF
DQKRATIQFHQPQRFKDELWRIQEKLECYF
GSLVGSNVYITPAGSQGLPPHYDDVEVFIL
QLEGEKHWRLYHPTVPLAREYSVEAEERIG
RPVHEFMLKPGDLLYFPRGTIHQADTPAGL
AHSTHVTISTYQNNSWGDFLLDTISGLVFD
TAKEDVELRTGIPRQLLLQVESTTVATRRL
SGFLRTLADRLEGTKELLSSDMKKDFIMHR
LPPYSAGDGAELSTPGGKLPRLDSVVRLQF
KDHIVLTVLPDQDQSDETQEKMVYIYHSLK
NSRETHMMGNEEETEFHGLRFPLSHLDALK
QIWNSPAISVKDLKLTTDEEKESLVLSLWT
ECLIQVV |
Tags and additions: N-terminal TEV cleavable 6His tag - mhhhhhhssgvdlgtenlyfq*s(m), cleaves at *. Tag removed: Yes
|
Host: E. coli BL21(DE3)-R3-pRARE2 |
Growth medium, induction protocol: Medium: TB + 50 µg/ml Kanamycin + 34 µg/ml chloramphenicol. 4 x 1 liter TB in 2.5-L baffled flasks were inoculated with 5 ml overnight culture and grown at 37°C. The protein expression was induced with 0.2 mM IPTG at OD600= 1.2 for 18 h at 18°C. |
Extraction buffer, extraction method: Lysis buffer: 50 mM HEPES pH 7.5, 500 mM NaCl, 20 mM imidazole, 0.5 mM TCEP, PMSF 0.5 mM and 15 units/ml Benzonase. Frozen cell pellets were thawed and resuspended in a total volume of 400 ml lysis buffer. The cells were disrupted by high pressure homogenisation at 15 kpsi (Avestin C5) . Cell debris were removed by centrifugation for 60 minutes at 40,000 x g. |
Column 1: 2 ml Ni-Sepharose 6 FF gravity column |
Column 1 Procedure: The column was equilibrated with 5 column 50 mM HEPES pH 7.5, 500 mM NaCl, 5% glycerol, 20 mM Imidazole, 0.5 mM TCEP. The lysate was applied to the column and allowed to flow through. The column was washed with 10 column volumes of 50 mM HEPES pH 7.5, 500 mM NaCl, 5% glycerol, 20 mM Imidazole, 0.5 mM TCEP, then more stringently with 50 mM HEPES pH 7.5, 500 mM NaCl, 5% glycerol, 40 mM Imidazole, 0.5 mM TCEP. The column was then eluted with 5 column volumes of 50 mM HEPES pH 7.5, 500 mM NaCl, 5% glycerol, 250 mM Imidazole, 0.5 mM TCEP. TEV protease was added to a final concentration of 60 ug/ml per mg of target protein, the digestion was allowed to proceed overnight at 4°C. |
Column 2: 10/20 HiPrep Desalting column |
Column 2 Procedure: The column was equilibrated with 50 mM HEPES pH 7.5, 50 mM NaCl. The digest was applied to the column and the buffer exchanged protein fraction collected. |
Column 3: 5 ml Q-Sepharose HP |
Column 3 Procedure: The column was equilibrated with 5 column volume 50 mM HEPES pH 7.5, 50 mM NaCl. The sample was applied to the column and the column eluted with a 20 column volume gradient from 50 mM to 500 mM NaCl. 1 ml fractions were collected and analysed by SDS-PAGE. The most pure fractions were pooled. |
Column 4: 10/20 HiPrep Desalting column |
Column 4 Procedure: The column was equilibrated with 10 mM HEPES pH 7.5, 500 mM NaCl, 5% glycerol and 0.5 mM TCEP. The sample was applied to the column and the buffer exchanged protein fraction collected. |
Concentration: The purified protein was concentrated to 11.5 mg/ml using an Amicon Ultracel ultrafiltration unit at 4,000 x g. Concentration was determined from absorbance at 280 nm using a NANODROP-1000 spectrophotometer. |
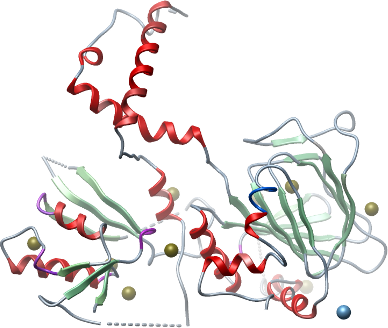
Crystallisation: Crystals were grown at 20°C by vapour diffusion in sitting drops by mixing protein (11.5 mg/ml) and well solution containing 12% PEG 3350; 0.005M CoCl2; 0.005M MgCl2; 0.005M CdCl2; 0.005M NiCl2; 0.1M HEPES pH 7.5 at a protein to precipitant ratio of 2:1. The crystal was cryo-protected using well solution supplemented with 20% (v/v) ethylene glycol and flash-cooled in liquid nitrogen. |
Data collection: Resolution: 2.6 Å; X-ray source: Synchrontron SLS-X10SA. |