Entry Clone Source: MGC |
Entry Clone Accession: IMAGE:3908548 |
SGC Construct ID: MUTA-c602 |
GenBank GI number: gi|4557767 |
Vector: pNIC-CTHF. Details [ PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
CTTAAGAAGGAGATATACTATGTCACCTCA
TTACCTGAGGCAGGTAAAAGAATCATCAGG
CTCCAGGCTCATACAGCAACGACTTCTACA
CCAGCAACAGCCCCTTCACCCAGAATGGGC
TGCCCTGGCTAAAAAGCAGCTGAAAGGCAA
AAACCCAGAAGACCTAATATGGCACACCCC
GGAAGGGATCTCTATAAAACCCTTGTATTC
CAAGAGAGATACTATGGACTTACCTGAAGA
ACTTCCAGGAGTGAAGCCATTCACACGTGG
ACCATATCCTACCATGTATACCTTTAGGCC
CTGGACCATCCGCCAGTATGCTGGTTTTAG
TACTGTGGAAGAAAGCAATAAGTTCTATAA
GGACAACATTAAGGCTGGTCAGCAGGGATT
ATCAGTTGCCTTTGATCTGGCGACACATCG
TGGCTATGATTCAGACAACCCTCGAGTTCG
TGGTGATGTTGGAATGGCTGGAGTTGCTAT
TGACACTGTGGAAGATACCAAAATTCTTTT
TGATGGAATTCCTTTAGAAAAAATGTCAGT
TTCCATGACTATGAATGGAGCAGTTATTCC
AGTTCTTGCAAATTTTATAGTAACTGGAGA
AGAACAAGGTGTACCTAAAGAGAAACTTAC
TGGTACCATCCAAAATGATATACTAAAGGA
ATTTATGGTTCGAAATACATACATTTTTCC
TCCAGAACCATCCATGAAAATTATTGCTGA
CATATTTGAATATACAGCAAAGCACATGCC
AAAATTTAATTCAATTTCAATTAGTGGATA
CCATATGCAGGAAGCAGGGGCTGATGCCAT
TCTGGAGCTGGCCTATACTTTAGCAGATGG
ATTGGAGTACTCTAGAACTGGACTCCAGGC
TGGCCTGACAATTGATGAATTTGCACCAAG
GTTGTCTTTCTTCTGGGGAATTGGAATGAA
TTTCTATATGGAAATAGCAAAGATGAGAGC
TGGTAGAAGACTCTGGGCTCACTTAATAGA
GAAAATGTTTCAGCCTAAAAACTCAAAATC
TCTTCTTCTAAGAGCACACTGTCAGACATC
TGGATGGTCACTTACTGAGCAGGATCCCTA
CAATAATATTGTCCGTACTGCAATAGAAGC
AATGGCAGCAGTATTTGGAGGGACTCAGTC
TTTGCACACAAATTCTTTTGATGAAGCTTT
GGGTTTGCCAACTGTGAAAAGTGCTCGAAT
TGCCAGGAACACACAAATCATCATTCAAGA
AGAATCTGGGATTCCCAAAGTGGCTGATCC
TTGGGGAGGTTCTTACATGATGGAATGTCT
CACAAATGATGTTTATGATGCTGCTTTAAA
GCTCATTAATGAAATTGAAGAAATGGGTGG
AATGGCCAAAGCTGTAGCTGAGGGAATACC
TAAACTTCGAATTGAAGAATGTGCTGCCCG
AAGACAAGCTAGAATAGATTCTGGTTCTGA
AGTAATTGTTGGAGTAAATAAGTACCAGTT
GGAAAAAGAAGACACTGTAGAAGTTCTGGC
AATTGATAATACTTCAGTGCGAAACAGGCA
GATTGAAAAACTTAAGAAGATCAAATCCAG
CAGGGATCAAGCTTTGGCTGAACGTTGTCT
TGCTGCACTAACCGAATGTGCTGCTAGCGG
AGATGGAAATATCCTGGCTCTTGCAGTGGA
TGCATCTCGGGCAAGATGTACAGTGGGAGA
AATCACAGATGCCCTGAAAAAGGTATTTGG
TGAACATAAAGCGAATGATCGAATGGTGAG
TGGAGCATATCGCCAGGAATTTGGAGAAAG
TAAAGAGATAACATCTGCTATCAAGAGGGT
TCATAAATTCATGGAACGTGAAGGTCGCAG
ACCTCGTCTTCTTGTAGCAAAAATGGGACA
AGATGGCCATGACAGAGGAGCAAAAGTTAT
TGCTACAGGATTTGCTGATCTTGGTTTTGA
TGTGGACATAGGCCCTCTTTTCCAGACTCC
TCGTGAAGTGGCCCAGCAGGCTGTGGATGC
GGATGTGCATGCTGTGGGCGTAAGCACCCT
CGCTGCTGGTCATAAAACCCTAGTTCCTGA
ACTCATCAAAGAACTTAACTCCCTTGGACG
GCCAGATATTCTTGTCATGTGTGGAGGGGT
GATACCACCTCAGGATTATGAATTTCTGTT
TGAAGTTGGTGTTTCCAATGTATTTGGTCC
TGGGACTCGAATTCCAAAGGCTGCCGTTCA
GGTGCTTGATGATATTGAGAAGTGTTTGGA
AAAGAAGCAGCAATCTGTAGCAGAGAACCT
CTACTTCCAATCGCACCATCATCACCACCA
TGATTACAAGGATGACGACGATAAGTGAGG
ATCC
|
Final protein sequence (tag sequence in lowercase)
MSPHYLRQVKESSGSRLIQQRLLHQQQPLH
PEWAALAKKQLKGKNPEDLIWHTPEGISIK
PLYSKRDTMDLPEELPGVKPFTRGPYPTMY
TFRPWTIRQYAGFSTVEESNKFYKDNIKAG
QQGLSVAFDLATHRGYDSDNPRVRGDVGMA
GVAIDTVEDTKILFDGIPLEKMSVSMTMNG
AVIPVLANFIVTGEEQGVPKEKLTGTIQND
ILKEFMVRNTYIFPPEPSMKIIADIFEYTA
KHMPKFNSISISGYHMQEAGADAILELAYT
LADGLEYSRTGLQAGLTIDEFAPRLSFFWG
IGMNFYMEIAKMRAGRRLWAHLIEKMFQPK
NSKSLLLRAHCQTSGWSLTEQDPYNNIVRT
AIEAMAAVFGGTQSLHTNSFDEALGLPTVK
SARIARNTQIIIQEESGIPKVADPWGGSYM
MECLTNDVYDAALKLINEIEEMGGMAKAVA
EGIPKLRIEECAARRQARIDSGSEVIVGVN
KYQLEKEDTVEVLAIDNTSVRNRQIEKLKK
IKSSRDQALAERCLAALTECAASGDGNILA
LAVDASRARCTVGEITDALKKVFGEHKAND
RMVSGAYRQEFGESKEITSAIKRVHKFMER
EGRRPRLLVAKMGQDGHDRGAKVIATGFAD
LGFDVDIGPLFQTPREVAQQAVDADVHAVG
VSTLAAGHKTLVPELIKELNSLGRPDILVM
CGGVIPPQDYEFLFEVGVSNVFGPGTRIPK
AAVQVLDDIEKCLEKKQQSVaenlyfq*sh
hhhhhdykddddk |
Tags and additions: C-terminal hexahistidine tag and Flag tag, TEV cleavable
|
Host: BL21(DE3)-R3-pRARE2 |
Expression:10µl of BL21(DE3)-R3 glycerol stock were inoculated into 5ml of Terrific broth medium supplemented with kanamycin (50 µg/ml) and chloramphenicol (34µg/ml) and grown overnight at 37°C, 200rpm. In the morning 1L of TB supplemented with the same antibiotics was inoculated with 10ml of the overnight culture and incubated at 37°C with intensive shaking (160rpm). After the OD600 reached 1.5, the temperature was changed to 18°C and IPTG was added to the final concentration of 1mM. The culture was incubated at 18°C with shaking (160rpm) for additional 18h. The following morning the 14 l culture was harvested and centrifuged for 10min at 4000rpm. Supernatant was discarded and the cell pellets were resuspended in 75ml of a lysis buffer and frozen at -80°C. |
Extraction: Lysis buffer: 50mM HEPES pH 7.5, 500mM NaCl, 5mM Imidazole, 5% glycerol + EDTA-free Complete (1 tablet/50ml).
The thawed cells were broken by 5 passes at 16.000 psi through a high pressure homogeniser, followed by centrifugation for 45 min at 20.000rpm at 4°C. |
Purification: Step 1: Ni-affinity, Ni-Sepharose - (GE Healthcare) Akta-Express; Step 2: Superdex 200 Column, HiPrep 16/60 (Amersham); Step 3: Ion exchange -5ml HiTrap Q Sepharose |
Buffers: Start buffer: 50mM HEPES pH 7.5, 500mM NaCl, 5mM Imidazole, 5% glycerol, 1mM PMSF, 0.5mM TCEP; Washing buffer: 50mM HEPES pH 7.5, 500mM NaCl, 10mM Imidazole, 5% glycerol, 1mM PMSF, 0.5mM TCEP; Elution buffer: 50mM HEPES pH 7.5, 500mM NaCl, 5% glycerol, 250mM Imidazole, 0.5mM TCEP. |
GF buffer: 10mM HEPES pH 7.5, 500mM NaCl, 5% glycerol, 0.5mM TCEP |
Procedure: The cell extract was loaded on the AKTA Express system. The extinction at 280nm was monitored and fractions were collected and analyzed by SDS-PAGE. MUTA containing fractions were diluted with IEX buffer A to a concentration of NaCl of 50 mM. The protein was loaded onto a HiTrap Q Sepharose column, and eluted with a NaCl gradient (50-300mM). Fractions containing protein were analysed by SDS-PAGE. |
Concentration and buffer exchange: Using Amicon Ultra-15 concentrators with 30kDa cutoff, the sample was concentrated to 11mg/ml. Concentrations were determined from the absorbance at 280 nm using a NanoDrop spectrophotometer. |
Mass spectrometry characterization: The calculated mass of the construct was 84724 Da, and the observed mass (ESI-MS) was 84510Da, compatible with an N-terminal methionine and serine deletion. |
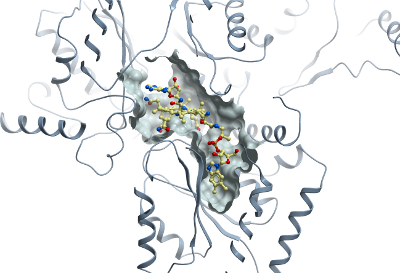
Crystallisation: Prior to crystallization protein (19.6 mg/ml) was pre-incubated with 250 µM adenosylcobalamin. Crystals were grown by vapour diffusion at 20°C, in 150 nl sitting drops mixing 100 nl protein and 50 nl mother liquor (30% PEG3350, 0.1 M Bis-Tris pH 5.5, 0.3 M NH4-sulphate) equilibrated against 20 µl reservoir containing mother liquor. Crystals were cryo-protected in mother liquor containing 25% (v/v) ethylene glycol and flash-cooled in liquid nitrogen. |
Data collection: Resolution: 1.95 Å; X-ray source: Swiss Light source (SLS), beamline X-10. |