Entry Clone Source: Synthetic |
Entry Clone Accession: n/a |
SGC Construct ID: YSK1A-c020 |
GenBank GI number: gi|21361358 |
Vector: pNIC-Zb. |
Amplified construct sequence:
ATGCACCATCATCATCATCATTCTTCTGGT
GTGGATAACAAGTTCAACAAGGAGCGTCGA
AGAGCTCGCCGTGAAATTCGCCATCTGCCG
AACCTGAACCGCGAACAGCGTCGCGCATTT
ATTCGCAGCCTGCGCGATGATCCGAGCCAG
AGCGCGAACCTGCTGGCGGAAGCGAAGAAG
CTGAACGATGCGCAGCCGAAGGGTACCGAG
AACCTGTACTTCCAATCCATGGCTCACCTC
CGGGGATTTGCCAACCAGCACTCTCGAGTG
GACCCTGAGGAGCTCTTCACCAAGCTCGAC
CGCATTGGCAAGGGCTCGTTTGGGGAGGTC
TACAAGGGCATCGATAACCACACAAAGGAG
GTGGTGGCCATCAAGATCATCGACCTGGAG
GAGGCCGAGGATGAGATCGAGGACATCCAG
CAGGAGATCACTGTCCTCAGTCAGTGCGAC
AGCCCCTACATCACCCGCTACTTTGGCTCC
TACCTAAAGAGCACCAAGCTATGGATCATC
ATGGAGTACCTGGGCGGCGGCTCAGCACTG
GACTTGCTTAAACCAGGTCCCCTGGAGGAG
ACATACATTGCCACGATCCTGCGGGAGATT
CTGAAGGGCCTGGATTATCTGCACTCCGAA
CGCAAGATCCACCGAGACATCAAAGCTGCC
AACGTGCTACTCTCGGAGCAGGGTGACGTG
AAGCTGGCGGACTTTGGGGTAGCAGGGCAG
CTCACAGACACGCAGATTAAGAGGAACACA
TTCGTGGGCACCCCCTTCTGGATGGCACCT
GAGGTCATCAAGCAGTCGGCCTACGACTTC
AAGGCTGACATCTGGTCCCTGGGGATCACA
GCCATCGAGCTGGCCAAGGGGGAGCCTCCA
AACTCTGACCTCCACCCCATGCGCGTCCTG
TTCCTGATTCCCAAGAACAGCCCACCCACA
CTGGAGGGCCAGCACAGCAAGCCCTTCAAG
GAGTTCGTGGAGGCCTGCCTCAACAAAGAC
CCCCGATTCCGGCCCACGGCCAAGGAGCTC
CTGAAGCACAAGTTCATCACACGCTACACC
AAGAAGACCTCCTTCCTCACGGAGCTCATC
GACCGCTATAAGCGCTGGAAGTCAGAGTGA
CAGTAAAGGTGGATACGGATCCGAATTCGA
GCTCCGTCGACAAGCTTGCGGCCGCACTCG
AGCACCACCACCACCACCACTGA
|
Final protein sequence (small letters refer to tag sequence remaining after TEF cleavage)
smAHLRGFANQHSRVDPEELFTKLDRIGKG
SFGEVYKGIDNHTKEVVAIKIIDLEEAEDE
IEDIQQEITVLSQCDSPYITRYFGSYLKST
KLWIIMEYLGGGSALDLLKPGPLEETYIAT
ILREILKGLDYLHSERKIHRDIKAANVLLS
EQGDVKLADFGVAGQLTDTQIKRNTFVGTP
FWMAPEVIKQSAYDFKADIWSLGITAIELA
KGEPPNSDLHPMRVLFLIPKNSPPTLEGQH
SKPFKEFVEACLNKDPRFRPTAKELLKHKF
ITRYTKKTSFLTELIDRYKRWKSE |
Tags and additions: *Cleavable N-terminal His6 and Z tag.
|
Host: BL21 (DE3) Rosetta-Phage resistant |
Expression protocol: Transformed 50 µl competent BL-21 (DE3) phage resistant cells with 10 µl of the plasmid DNA and plated out onto LB plate plus 50 µg/ml kanamycin and 35 µg/ml chloramphenicol. The next day colonies were picked into fresh deep well blocks containing 1 ml LB + 50 µg/ml kanamycin and 35 µg/ml chloramphenicol. Cultures were grown overnight and glycerol stocks were prepared by adding 333 ml of 60 % glycerol to 1 ml of cell suspension, which were stored at -80°C and used for future scale up preparations.
The glycerol stock was used to inoculate 10 ml of LB supplemented with 50 µg/ml kanamycin and 35 µg/ml chloramphenicol. This starter culture was grown overnight at 37°C and used to inoculate a 1 liter culture in the same medium. The culture was grown at 37°C until the OD600 reached ~0.5. After that the temperature was lowered to 18°C. Protein production was induced with 1mM IPTG and recombinant YSK1 was expressed at that temperature over night. The next day cells were harvested by centrifugation at 4000 rpm for 15 minutes. The cell pellet was stored at -80°C degrees. |
Lysis and Ni-affinity chromatography:
Buffers: Binding buffer: 50 mM HEPES pH 7.5, 300mM NaCl,, 20 mM Imidazole. Wash buffer 1: 50 mM HEPES pH 7.5, 1M NaCl, 20mM Imidazole.Wash buffer 2: as for lysis buffer. Elution buffer: 50mM HEPES pH 7.5, 300mM NaCl, 150 mM Imidazole.
Procedure: The cell pellet form a two litr culture was re-suspended in one volume (25 ml) of binding buffer and the re-suspended cells were lysed by sonication. The lysate was cleared by a centrifugation at 20,000 rpm (4°C).
5 ml of 50% Ni-NTA slurry (Qiagen) was applied to a 1.5 x 10 cm gravity column. The column was equilibrated with 100 ml binding buffer. The lysate was applied to the column and was subsequently washed with 50 ml wash buffer 1 and 2. YSK1 was eluted with 25 mls of elution buffer. The eluted protein was collected and analyzed by SDS-PAGE. DTT was added to the protein sample to a final concentration of 10mM. The his-tag was removed by the addition of 100 mg of Tev protease and the eluted protein sample was incubated overnight at 4°C. |
SEC-Buffers: Column 2: Size exclusion chromatography (Superdex S200, 60 x 1cm)
SEC-Buffers: 50 mM Hepes, pH 7.5, 300 mM NaCl, 5 mM DTT.
Procedure: The fractions eluted of the Ni-affinity chromatography were concentrated to about 4 mls using Centricon concentrators (10kDa cut off). The concentrated protein was applied to a Superdex S200 column equilibrated in SEC buffer at a flow rate of 0.8 ml/min. Eluted fractions were 95% pure as judged by SDS-PAGE. |
Mass spectrometry characterization: ESI-MS revealed that the the TEV cleaved protein had the mass of 33750 Da which is the expected mass with the addition of 2 phosphorylation adducts. |
Protein concentration: 11.3 mg/ml in SEC buffer using a centricon with a 10kDa cut off |
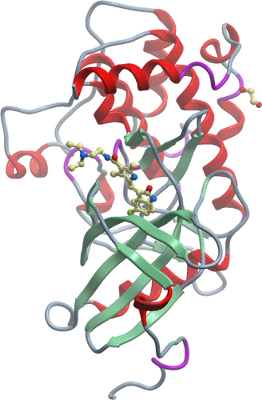
Crystallisation: YSK1 was crystallized at 4 °C using the sitting-drop vapor diffusion method at 11.3 mg/ml in the presence of 1 mM of inhibitor K00239b (CalBioChem: #572660) added from a 50 mM DMSO stock. Diffraction quality crystals were obtained by mixing 50 nl of protein solution with 150 nl of crystallization buffer 0.2M potassium citrate, 0.1M BisTrisPropane pH 8.5, 20% PEG3350 and 10% Ethylene glycol. |
Data collection: Resolution: Crystals were flash frozen in liquid nitrogen after being cryo protected in 30% propylene glycole (w/v) in the well solution. |