Entry Clone Source: Origene |
Entry Clone Accession: NM_015061 variant |
SGC Construct ID: JMJD2CA-c604 |
GenBank GI number: gi|24307987 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
CATATGCACCATCATCATCATCATTCTTCT
GGTGTAGATCTGGGTACCGAGAACCTGTAC
TTCCAATCCATGGAGGTGGCCGAGGTGGAA
AGTCCTCTGAACCCCAGCTGTAAGATAATG
ACCTTCAGACCCTCCATGGAGGAGTTCCGG
GAGTTCAACAAATACCTTGCATACATGGAG
TCTAAAGGAGCCCATCGTGCGGGTCTTGCA
AAGGTGATTCCTCCTAAGGAGTGGAAGCCA
AGACAGTGCTATGATGACATTGATAATTTG
CTCATTCCAGCACCAATTCAGCAGATGGTC
ACAGGGCAGTCAGGACTGTTCACTCAGTAC
AACATCCAGAAAAAAGCGATGACTGTGAAG
GAGTTCAGGCAGCTGGCCAACAGTGGCAAA
TATTGTACTCCAAGATACTTGGATTACGAA
GATTTGGAGCGCAAGTACTGGAAGAACTTA
ACTTTTGTGGCACCTATCTATGGTGCAGAT
ATTAATGGGAGCATATATGATGAGGGTGTG
GATGAATGGAACATAGCTCGCCTCAATACA
GTCTTGGATGTGGTTGAAGAAGAGTGTGGC
ATTTCTATTGAGGGTGTAAATACCCCATAT
CTCTATTTTGGCATGTGGAAGACCACGTTT
GCATGGCACACCGAAGACATGGACCTCTAT
AGCATTAATTATCTCCACTTTGGAGAGCCC
AAGTCTTGGTATGCTATACCTCCGGAGCAT
GGAAAACGACTTGAAAGACTAGCTCAAGGT
TTTTTCCCAAGCAGCTCCCAAGGGTGTGAT
GCATTTCTTCGCCACAAGATGACATTGATT
TCTCCATCAGTATTGAAGAAATATGGTATT
CCCTTTGACAAGATAACCCAGGAGGCTGGA
GAATTCATGATCACTTTCCCATATGGCTAC
CATGCTGGTTTTAATCATGGTTTCAACTGT
GCAGAATCTACAAATTTTGCTACTGTCAGA
TGGATTGACTATGGAAAAGTTGCCAAATTG
TGCACTTGCAGGAAAGACATGGTGAAGATT
TCAATGGATATCTTTGTGAGGAAATTTCAG
CCAGACAGATATCAGCTTTGGAAACAAGGA
AAGGATATATACACCATTGATCACACGAAG
TGACAGTAAAGGTGGATACGGATCCGAA |
Final protein sequence
SMEVAEVESPLNPSCKIMTFRPSMEEFREF
NKYLAYMESKGAHRAGLAKVIPPKEWKPRQ
CYDDIDNLLIPAPIQQMVTGQSGLFTQYNI
QKKAMTVKEFRQLANSGKYCTPRYLDYEDL
ERKYWKNLTFVAPIYGADINGSIYDEGVDE
WNIARLNTVLDVVEEECGISIEGVNTPYLY
FGMWKTTFAWHTEDMDLYSINYLHFGEPKS
WYAIPPEHGKRLERLAQGFFPSSSQGCDAF
LRHKMTLISPSVLKKYGIPFDKITQEAGEF
MITFPYGYHAGFNHGFNCAESTNFATVRWI
DYGKVAKLCTCRKDMVKISMDIFVRKFQPD
RYQLWKQGKDIYTIDHTK |
Tags and additions: N-terminal, TEV cleavable hexahistidine tag |
Expression - JMJD2CA catalytic domain
Expression strain: BL-21(DE3)-R3-Rosetta (A homemade phage resistant version of BL21(DE3) containing the pRARE2 plasmid from Rosetta II (DE3) cells).
Transformation: The construct DNA was transformed into competent cells of the expression strain by a standard heat shock procedure.
Glycerol stock preparation: A number of colonies were used to inoculate 1 ml of LB media containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol, which was incubated in a 37°C shaker overnight. The next day glycerol stocks were prepared from this overnight culture.
Expression: 2 ml of a glycerol stock was used to inoculate 40 ml of LB containing 50 µg/ml kanamycin and 34 µg/ml chloramphenicol, which was incubated at 37°C overnight.
15ml starter culture was used per litre TB, containing 50 µg/ml kanamycin. The culture was incubated at 37°C until OD~1, when the temperature of the incubator was reduced to 18°C. Expression was induced with 0.1 mM IPTG and the culture continued o/n.
Cell harvest: Cells were pelleted at 6238x g for 15 min at 4°C, and stored at -80°C. The yield was 10g cells / litre culture.
|
Purification - JMJD2CA catalytic domain
Cell Lysis: Lysis Buffer: 50mM HEPES pH 7.4, 500mM NaCl, 5% glycerol, 20 mM Imidazole pH 7.4, 0.5 mM TCEP, 1mM PMSF
The pellets were resuspended in lysis buffer. They were passed 4 times through an Emulsiflex C5 high-pressure homogeniser, collecting a final volume of approximately 35 ml/ litre culture. Cell debris and DNA were spun down at 45000x g, 60 min ( Beckman JA 18 17500 rpm). The supernatant was collected to which 12.5 U of Benzonase was added with a 30-60 min incubation on ice.
Purification:
Wash Buffer: 50 mM Hepes pH 7.4, 500 mM NaCl, 5% Glycerol, 70 mM Imidazole pH 7.4, 0.5 mM TCEP
Elution Buffer: 50 mM Hepes pH 7.4, 500 mM NaCl, 5% Glycerol, 250 mM Imidazole pH 7.4, 0.5 mM TCEP
Gel Filtration buffer: 50 mM Hepes pH 7.4, 500 mM NaCl , 5% glycerol, 0.5 mM TCEP
Purification was performed in an Akta Express system (GE Healthcare) with an automated program for Ni-affinity (HisTrap FF) and gel filtration (HiLoad 16/60 S200) chromatography steps.
Step 1: Ni-affinity, HisTrap Crude FF, 1 ml
The clarified cell extract was loaded on the column at 0.8 ml/min on the AKTA-express system. The column was washed with 40 cv of lysis buffer, 10 cv of wash buffer, and then eluted with elution buffer at 0.8 ml/min. The eluted peak of A280 was automatically collected.
Step 2: Gel filtration, Hiload 16/60 Superdex S200 prep grade, 120 ml
The eluted fractions from the Ni-affinity column was loaded on the gel filtration column pre-equilibrated in Gel Filtration buffer at 1.2 ml/min. Eluted proteins were collected in 1.8 ml fractions and analyzed on SDS-PAGE.
|
TEV protease digestion and concentration -JMJD2CA catalytic domain
TEV protease digestion: Peak fractions from the gel filtration containing JMJD2CA were pooled and TEV protease was added at a molar ratio of 1:22. The digestion was left overnight at 4 °C.
SDS-PAGE and Mass Spec confirmed TEV digestion. His-TEV and contaminating proteins were removed by binding to Ni resin, pre-equilibrated in GF buffer. The flow through containing TEV-cleaved protein, was collected and concentrated using a Amicon centrifugal filter with a 30kDa MWCO.
To remove any precipitation, the concentrated protein was centrifuged 20 min/14000rpm/4°C and the supernatant was collected. The final concentration was 7 mg protein /ml and yield 1.5 mg/ litre culture. The protein was flash frozen and stored at -80°C in 70µl aliquots.
|
Mass spectrometry characterisation: Measured: 40416.1; Expected: 40415.4 |
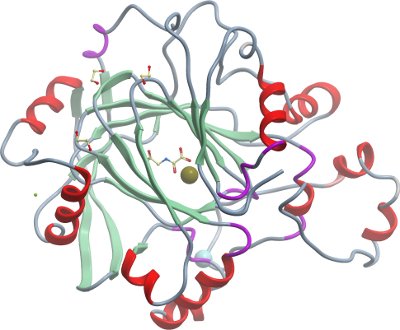
Crystallisation:
Prior to crystallization, protein was supplemented with 2mM
N-oxalylglycine (NOG). Crystals were grown at 4°C by vapour diffusion in
sitting drops mixing protein (7 mg/ml) and well solution (25% PEG3350,
0.2M sodium nitrate, 0.1M Bis-Tris propane pH 6.5, 5% ethylene glycol,
0.01M NiCl2) at a ratio of 2:1. Crystals were cryo-protected using well
solution supplemented with 25% (v/v) ethylene glycol and flash-cooled in
liquid nitrogen. |
Data collection: Resolution: 2.55 Å; X-ray source: Diamond Light Source beamline I24 |