Entry Clone Source: MGC |
Entry Clone Accession: IMAGE:4243419 |
SGC Construct ID: TTC5A-c022 |
GenBank GI number: gi|24308432 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
TTACTTCCAATCCATGCGGCAACGAG
AGCAACAACTTCTGGAATTCCTGGAT
AGATTAACCAGCCTCCTTGAGAGTAA
GGGAAAGGTGAAGACCAAAAAGCTGC
AGAGCATGCTGGGAAGCTTGCGCCCA
GCCCATCTAGGCCCTTGCAGTGATGG
GCACTATCAGTCAGCCTCTGGGCAGA
AAGTGACCCTGGAGCTCAAGCCACTG
AGTACGCTTCAGCCTGGGGTGAACAG
CGGTGCCGTCATCCTGGGAAAGGTGG
TATTTAGCCTCACCACAGAGGAGAAA
GTCCCCTTTACATTTGGCCTGGTAGA
TTCAGATGGACCTTGCTATGCAGTGA
TGGTGTACAATATAGTGCAGAGCTGG
GGAGTGCTCATTGGAGACTCTGTAGC
CATTCCTGAGCCCAACCTGCGGCTTC
ACCGAATTCAGCACAAAGGAAAGGAC
TATTCCTTTTCCAGTGTTCGAGTGGA
GACGCCCCTCCTGCTAGTGGTGAATG
GGAAGCCTCAGGGATGACAGTAAAGG
TGGATA
|
Final protein sequence (Tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfq^sMRQ
REQQLLEFLDRLTSLLESKGKVKTKK
LQSMLGSLRPAHLGPCSDGHYQSASG
QKVTLELKPLSTLQPGVNSGAVILGK
VVFSLTTEEKVPFTFGLVDSDGPCYA
VMVYNIVQSWGVLIGDSVAIPEPNLR
LHRIQHKGKDYSFSSVRVETPLLLVV
NGKPQG
^ TEV cleave site |
Tags and additions: mhhhhhhssgvdlgtenlyfq^s(m) TEV-cleavable (^) N-terminal his6 tag. |
Host: BL21 (DE3) R3-pRARE2.
|
Growth medium, induction protocol: A glycerol stock was used to inoculate a 50ml starter culture containing LB media with 50µg/ml Kanamycin and 34µg/ml chloramphenicol. The starter culture was grown overnight at 37°C with shaking at 200 rpm. The following morning, four flasks containing 1 L TB/kanamycin/chloramphenicol were each inoculated with 5 ml of the starter culture. Cultures were incubated at 37°C with shaking at 180 rpm until an OD600nm ? 0.6 was reached. The flasks were then cooled down to 18°C and 0.3mM IPTG added to induce protein expression overnight. Cells were harvested by centrifugation at 4500 rpm at 4°C for 15 min.
Binding buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole
Extraction buffer, extraction method: Cell pellets were resuspended in 80ml binding buffer supplemented with 0.5 mM TCEP and a 1:2000 dilution of Calbiochem protease inhibitor SET V. The cells were lysed by ultrasonication over 10 min with the sonicator pulsing ON for 5 sec and OFF for 10 sec. To precipitate nucleic acids, PEI (polyethyleneimine) was added to the lysate to a final concentration of 0.15% and the lysate was centrifuged at 16,500 rpm for 40 minutes. The supernatant was collected for purification and passed through a 0.8µm filter. |
Column 1: Ni-Affinity Chromatography. 3ml of 50% slurry in 1.5 x 10cm column, washed with binding buffer. |
Column 1 Buffer:
Binding buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 5mM Imidazole
Wash buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 30mM Imidazole
Elution buffer: 50mM HEPES, pH 7.5; 500mM NaCl; 5% Glycerol; 50 to 250mM Imidazole |
Column 1 Procedure: The supernatant was applied by gravity flow onto the Ni-sepharose column. The column was then washed with ~10 CV (column volume) binding buffer and ~20 CV wash buffer. The bound protein was eluted by applying a step gradient of imidazole (6ml fractions of elution buffer sequentially containing 50mM, 100mM, 150mM or 250mM imidazole). 10mM DTT was added to each fraction collected for overnight storage at 4°C. |
Enzymatic treatment: 100µl TEV protease (2mg/ml) was added for overnight cleavage of the N-terminal hexahistidine tag. |
Column 2: Size Exclusion Chromatography - S200 HiLoad 16/60 Superdex run on ÄKTA-Express. |
Column 2 Buffers:
Gel Filtration buffer: 50 mM HEPES pH 7.5, 250 mM NaCl.
|
Column 2 Procedure: Prior to applying the protein, the S200 16/60 column was washed and equilibrated with gel filtration buffer. The 250 mM imidazole elution fraction was the cleanest fraction from the Ni-sepharose column and was concentrated to 3 ml using a 5 kD MWCO Amicon Ultra concentrator. The concentrated protein was directly applied onto the equilibrated S200 16/60 column, and run at a flow-rate of 1 ml/min. Fractions containing the protein were pooled together, and 10mM DTT was added for overnight storage at 4°C. The main peak eluted at 98ml. |
Column 3: Ni-Affinity Chromatography (Ni-rebinding). 0.5ml of 50% slurry in 1.5 x 10cm column, washed with binding buffer. |
Column 3 Buffer:
Binding buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% Glycerol; 5 mM Imidazole
Wash buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% Glycerol; 30 mM Imidazole
Elution buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% Glycerol; 250 mM Imidazole |
Column 3 Procedure: Eluted TTC5 protein from gel filtration was pooled and supplemented with 10 mM imidazole. The protein was applied by gravity flow onto the column and collected in the flow through. The column was then washed with 10 ml wash buffer. Remaining contaminant protein (e.g. TEV protease) was then eluted by applying a step gradient of imidazole (6 ml fractions of elution buffer sequentially containing 50mM, 100mM, 150mM or 250mM imidazole). All TTC5 protein was recovered in the flow through. |
Mass spectrometry characterization: The purified native protein was homogeneous and had an experimental mass of 18,159.6 Da (expected MW = 18159.1). Masses were determined by LC-MS, using an Agilent LC/MSD TOF system with reversed-phase HPLC coupled to electrospray ionisation and an orthogonal time-of-flight mass analyser. |
Protein concentration: 0.5 mM TCEP was added to the sample and the protein was concentrated in a 5 kD MWCO Amicon Ultra concentrator to 3.5 mg/ml using an estimated extinction coefficient of 11460 and MW of 18159. |
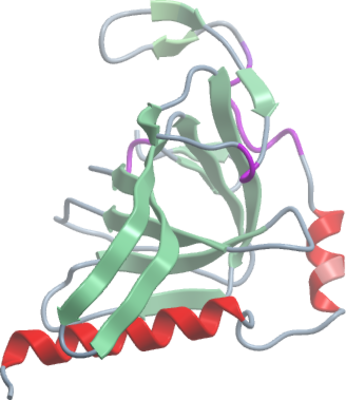
Crystallisation: Protein at 3.5 mg/ml was buffered in 50 mM HEPES, pH 7.5, 250 mM NaCl, 0.5 mM TCEP. Clusters of thin needles grew from a number of PEG/salt-containing conditions. The best crystals were grown from drops containing equal volumes of protein (3.5mg/ml) and reservoir solution containing 36µl of 0.2M sodium iodide, 20% PEG3350, 10% ethylene glycol mixed with 4µl of 0.2M sodium acetate, 20% PEG3350, 10% ethylene glycol, 0.1M bis-tris propane pH8.5. Crystals belong to space group P212121 and contain a single molecule per ASU. All crystals were vitrified in well solution supplemented with 25-30% ethylene glycol. |
Data Collection: The native crystal diffracted to a resolution of 1.8 Å
X-ray source: Swiss Light Soucre beamline X10SA. |