Entry Clone Source: Synthetic |
Entry Clone Accession: n/a |
SGC Construct ID: JMJD3A-c023 |
GenBank GI number: gi|89041169 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
CATATGCACCATCATCATCATCATTC
TTCTGGTGTAGATCTGGGTACCGAGA
ACCTGTACTTCCAATCCATGCTGCCC
CGGGAAAAACTCAACCCCCCTACACC
CAGCATCTATCTGGAGAGCAAACGGG
ATGCCTTCTCACCTGTCCTGCTGCAG
TTCTGTACAGACCCTCGAAATCCCAT
CACAGTGATCCGGGGCCTGGCGGGCT
CCCTGCGGCTCAACTTGGGCCTCTTC
TCCACCAAGACCCTGGTGGAAGCGAG
TGGCGAACACACCGTGGAAGTTCGCA
CCCAGGTGCAGCAGCCCTCAGATGAG
AACTGGGATCTGACAGGCACTCGGCA
GATCTGGCCTTGTGAGAGCTCCCGTT
CCCACACCACCATTGCCAAGTACGCA
CAGTACCAGGCCTCATCCTTCCAGGA
GTCTCTGCAGGAGGAGAAGGAGAGTG
AGGATGAGGAGTCAGAGGAGCCAGAC
AGCACCACTGGAACCCCTCCTAGCAG
CGCACCAGACCCGAAGAACCATCACA
TCATCAAGTTTGGCACCAACATCGAC
TTGTCTGATGCTAAGCGGTGGAAGCC
CCAGCTGCAGGAGCTGCTGAAGCTGC
CCGCCTTCATGCGGGTAACATCCACG
GGCAACATGCTGAGCCACGTGGGCCA
CACCATCCTGGGCATGAACACGGTGC
AGCTGTACATGAAGGTGCCCGGCAGC
CGAACGCCAGGCCACCAGGAGAATAA
CAACTTCTGCTCCGTCAACATCAACA
TTGGCCCAGGCGACTGCGAGTGGTTC
GCGGTGCACGAGCACTACTGGGAGAC
CATCAGCGCTTTCTGTGATCGGCACG
GCGTGGACTACTTGACGGGTTCCTGG
TGGCCAATCCTGGATGATCTCTATGC
ATCCAATATTCCTGTGTACCGCTTCG
TGCAGCGACCCGGAGACCTCGTGTGG
ATTAATGCGGGGACTGTGCACTGGGT
GCAGGCCACCGGCTGGTGCAACAACA
TTGCCTGGAACGTGGGGCCCCTCACC
GCCTATCAGTACCAGCTGGCCCTGGA
ACGATACGAGTGGAATGAGGTGTGAC
AGTAAAGGTGGATACGGATCCGAA
|
Final protein sequence (Tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfq^smLP
REKLNPPTPSIYLESKRDAFSPVLLQ
FCTDPRNPITVIRGLAGSLRLNLGLF
STKTLVEASGEHTVEVRTQVQQPSDE
NWDLTGTRQIWPCESSRSHTTIAKYA
QYQASSFQESLQEEKESEDEESEEPD
STTGTPPSSAPDPKNHHIIKFGTNID
LSDAKRWKPQLQELLKLPAFMRVTST
GNMLSHVGHTILGMNTVQLYMKVPGS
RTPGHQENNNFCSVNINIGPGDCEWF
AVHEHYWETISAFCDRHGVDYLTGSW
WPILDDLYASNIPVYRFVQRPGDLVW
INAGTVHWVQATGWCNNIAWNVGPLT
AYQYQLALERYEWNEV
^ TEV cleavage site |
Tags and additions: N-terminal TEV cleavable 6His tag. |
Host: BL21 (DE3) slyD-.
|
Growth medium, induction protocol: TB + 50µg/ml kanamycin + 34µg/ml chloramphenicol. 30 x 1 litre TB in 2.5L baffled flasks were inoculated with 30ml overnight culture and grown at 37°C. The protein expression was induced with 0.2 mM IPTG at OD600 = 0.8 for 18 hours at 18°C. The cells were collected by centrifugation and frozen at -80°C.
Lysis buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 20 mM Imidazole; PMSF 1 mM; 15 units/ml Benzonase.
Extraction buffer, extraction method: Frozen cell pellets were thawed and resuspended in a total volume of 800ml lysis buffer. The cells were disrupted by high pressure homogenisation (15kpsi) followed by sonication. Cell debris were removed by centrifugation for 60 minutes at 30000g. |
Column 1: Ni-affinity, HisTrap FF Crude, 5ml (GE/Amersham Biosciences) |
Column 1 Buffer:
Wash Buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 40 mM Imidazole.
Elution Buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 250 mM Imidazole.
|
Column 1 Procedure: The cell extract was loaded on the column at 5ml/min using a persitaltic pump. The column was then washed with 10 volumes of lysis buffer, 10 volumes of wash buffer, and then eluted with elution buffer. The eluted peak of A280 was automatically collected. |
Column 2: 5ml HisTrap Q Sepharose High Performance (GE/Amersham Biosciences). |
Column 2 Buffers:
IEX Binding Buffer: 10 mM HEPES, pH 7.5; 50 mM NaCl; 1 mM DTT.
IEX Elution Buffer: 10 mM HEPES, pH 7.5; 1 M NaCl; 1 mM DTT. |
Column 2 Procedure: The eluted fractions from the Ni-Affinity HisTrap column were TEV-cleaved overnight at 4°C. The protein was buffer-exchanged into IEX binding buffer, and loaded onto the column. The elution is achieved by linear NaCl gradient with IEX elution buffer. Fractioons containing JMJD3 were pooled. |
Column 3: HiLoad 26/60 Superdex 200 (GE/Amersham Biosciences). |
Column 3 Buffer: 10 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 0.5 mM TCEP. |
Column 3 Procedure: The protein pooled from the ion exchange step was applied to the column pre-equilibrated with GF buffer, and fractions were collected at 1.0ml/min |
Protein concentration: The protein was concentrated using an Amicon centrifugal concentrator (10kDa MWCO) to 15mg/ml as determined by A280. |
Mass spectrometry characterization: The mass determined for JMJD3A-p023 was 37650 Da, in agreement with the predicted mass for the His-tagged protein. |
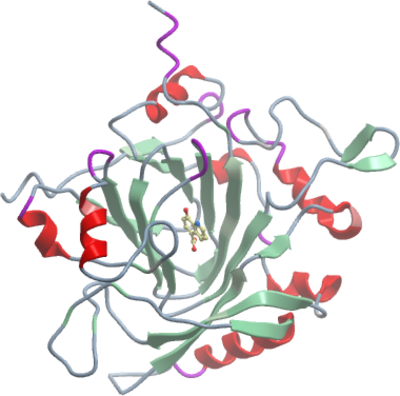
Crystallisation: Crystals were grown by vapour diffusion at 4°C. A sitting drop consisting of 100µl protein (11mg/ml) supplemented 1 mM 8-hydroxy-5-caryboxyquinoline and 50µl well solution was equilibrated against well solution containing 0.2 M MgCl2, 0.1 M HEPES pH7.5, 25% (v/v) PEG 3350, 2 mM NiCl2. Crystals were cryoprotected with well solution supplemented with 25% (v/v) ethylene glycol and 5 mM 8-hydroxy-5-carboxyquinoline, and flash frozen in liquid nitrogen. |
Data Collection: 1.80Å
X-ray source: Diamond Light Source beamline I02. |