Entry Clone Source: MGC |
Entry Clone Accession: IMAGE:4825802 |
SGC Construct ID: ANLNA-c002 |
GenBank GI number: gi|31657094 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
ATGCACCATCATCATCATCATTCTTC
TGGTGTAGATCTGGGTACCGAGAACC
TGTACTTCCAATCCATGAAATGTCAA
GTGAATTCCAGTGTTGAAGAAAGAGG
TTTTCTAACCATATTTGAAGATGTTA
GTGGTTTTGGTGCCTGGCATCGAAGA
TGGTGTGTTCTTTCTGGAAACTGTAT
ATCTTATTGGACTTATCCAGATGATG
AGAAACGCAAGAATCCCATAGGAAGG
ATAAATCTGGCTAATTGTACCAGTCG
TCAGATAGAACCAGCCAACAGAGAAT
TTTGTGCAAGACGCAACACTTTTGAA
TTAATTACTGTCCGACCACAAAGAGA
AGATGACCGAGAGACTCTTGTCAGCC
AATGCAGGGACACACTCTGTGTTACC
AAGAACTGGCTGTCTGCAGATACTAA
AGAAGAGCGGGATCTCTGGATGCAAA
AACTCAATCAAGTTCTTGTTGATATT
CGCCTCTGGCAACCTGATGCTTGCTA
CAAATGA
|
Final protein sequence (Tag sequence in lowercase):
smKCQVNSSVEERGFLTIFEDVSGFG
AWHRRWCVLSGNCISYWTYPDDEKRK
NPIGRINLANCTSRQIEPANREFCAR
RNTFELITVRPQREDDRETLVSQCRD
TLCVTKNWLSADTKEERDLWMQKLNQ
VLVDIRLWQPDACYK
(Lys977 to Lys1119)
The N-terminal residues, sm, derive from the vector following TEV protease digestion to remove the expression tag. |
Tags and additions: N-terminal, TEV cleavable hexahistidine tag. |
Host: BL21 (DE3)R3-pRARE2 (Phage resistant strain).
|
Growth medium, induction protocol: The construct DNA was transformed into homemade chemically competent cells of the expression strain by a standard heat shock procedure. A number of colonies from the transformation were used to inoculate 1ml of LB media containing 50µg/ml kanamycin and 34µg/ml chloramphenicol, which was placed in a 37°C shaker overnight. The next day glycerol stocks were prepared from this overnight culture. A glycerol stock was used to inoculate 40ml of LB media containing 50µg/ml kanamycin and 34µg/ml chloramphenicol, which was placed in a 37°C shaker overnight. The next day 2x15ml of this starter culture was used to inoculate 2x1L of LB media containing 40µg/ml kanamycin in 2x2L baffled shaker flasks. When the OD600 was approximately 1.0, the temperature was reduced to 18°C and the cells were induced by the addition of 0.5mM IPTG. The expression was continued overnight. Cells were spun at 5500rpm for 10 mins and the pellets resuspended in Lysis Buffer and then frozen at -20°C.
Lysis buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% Glycerol; 5 mM Imidazole; 0.5 mM TCEP.
Extraction buffer, extraction method: The resuspended cell pellet was thawed and lysed by sonication. The sample was diluted to 90ml volume and PEI (polyethyleneimine) was added to a final concentration of 0.15%. The cell debris and precipitated DNA were spun down (18000rpm, JA18 rotor, 60 min). |
Column 1: 2ml of Ni-NTA in a 1cm diameter gravity flow column. |
Column 1 Buffers:
Binding Buffer:
50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 5 mM Imidazole; 0.5 mM TCEP.
Wash Buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 25 mM Imidazole; 0.5 mM TCEP.
Elution Buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 250 mM Imidazole; 0.5 mM TCEP. |
Column 1 Procedure: The clarified supernatant was passed through the column. The column was washed with 50ml of binding Buffer and 25ml of Wash Buffer. 10ml of Elution Buffer was passed through to elute the protein. |
Column 2: S200 16/60 Gel Filtration |
Column 2 Buffer: 25 mM HEPES, pH 7.5; 150 mM NaCl; 0.5 mM TCEP. |
Column 2 Procedure: The eluted protein was concentrated to 5ml volume and injected onto an S200 16/60 GF column (pre-equilibrated in GF buffer) at 1.0 ml/min. 1.75ml fractions were collected. |
Enzymatic treatment: The gel filtration fractions containing ANLNA were pooled. TEV protease was added and the sample left overnight at 4°C. The digested sample was passed through a 0.5cm diameter column containing 2ml of Ni-NTA (pre-equilibrated in GF Buffer). The resin was eluted with a further 10ml of GF Buffer containing 10 mM Imidazole. |
Protein concentration: The sample was concentrated to 19mg/ml (measured by 280nm absorbance), distributed into aliquots and frozen at -80°C. |
Mass spectrometry characterization:
Measured: 17220 (+80)
Expected: 17140.5
|
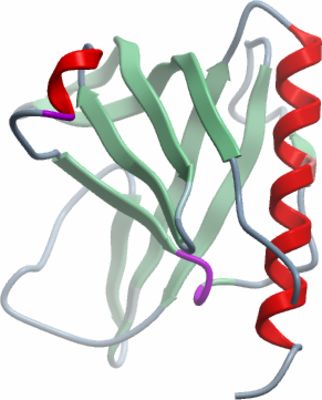
Crystallisation: Crystals grew from a 2:1 ratio of 6mg/ml protein and precipitant solution (20% PEG3350, 0.1M BisTris pH 5.5, 4% acetonitrile), using the vapour diffusion method. Seeds from crushed ANLNA crystals were added to enhance crystal size. |
Data collection: Crystals were cryo-protected by equilibration into precipitant solution containing 25% ethylene glycol, and then flash frozen in liquid nitrogen. Data was collected at Diamond beamline I02. |