Entry Clone Source: Site directed mutagenesis |
Entry Clone Accession: IMAGE:3357140 |
SGC Construct ID: MLYCDA-c506 |
GenBank GI number: gi|6912498 |
Vector: pNIC28-Bsa4. Details [PDF ]; Sequence [ FASTA ] or [ GenBank ] |
Amplified construct sequence:
CATATGCACCATCATCATCATCATTC
TTCTGGTGTAGATCTGGGTACCGAGA
ACCTGTACTTCCAATCCATGGACGAG
CTGCTGCGCCGCGCGGTGCCGCCGAC
GCCGGCCTACGAGCTGCGCGCAGCAA
CACCGGCGCCCGCCGAGGGTCAGTGC
GCGGACTTCGTGAGCTTCTACGGTGG
GCTGGCCGAGACGGCCCAGCGGGCCG
AACTGCTGGGCCGCCTGGCGCGGGGC
TTCGGCGTGGACCACGGCCAGGTGGC
GGAGCAGAGCGCCGGCGTGCTCCATC
TGCGCCAGCAGCAGCGGGAGGCGGCG
GTGCTGCTGCAGGCCGAGGACCGGCT
GCGCTACGCGCTGGTGCCGCGCTATC
GCGGCCTCTTCCACCACATCAGCAAG
CTGGACGGCGGCGTGCGCTTCCTGGT
GCAGCTGCGGGCCGACCTGCTGGAGG
CGCAGGCCCTCAAGCTGGTGGAGGGG
CCGGACGTCCGGGAAATGAATGGGGT
GCTGAAAGGAATGCTCTCAGAATGGT
TTTCCCCCGGGTTCCTGAACCTAGAA
CGGGTTACCTGGCATTCACCGTGTGA
AGTGCTTCAGAAAATCAGTGAGGCTG
AGGCTGTGCATCCTGTAAAAAACTGG
ATGGACATGAAGCGCCGCGTTGGGCC
CTACAGAAGGTGTTACTTCTTTTCTC
ACTGTTCGACCCCTGGGGAGCCCCTG
GTCGTTTTGCACGTGGCACTGACTGG
TGACATCTCCAGCAACATCCAGGCAA
TCGTGAAGGAACATCCTCCATCAGAA
ACAGCAGCTGCTAACAAAATCACTGC
TGCGATCTTTTATTCCATCAGCTTGA
CCCAGCAGGGACTCCAAGGGGTGGAG
CTGGGAACATTCCTCATAAAGCGAGT
CGTCAAGGAGTTGCAGAGAGAGTTTC
CTCACCTTGGGGTGTTTTCAAGTCTG
TCACCTATACCTGGTTTCACCAAATG
GCTTCTGGGGCTTCTGAACTCGCAAA
CGAAGGAGCATGGGAGGAATGAACTC
TTTACAGATTCGGAATGTAAGGAAAT
CTCGGAGATCACAGGTGGCCCCATTA
ACGAGACCCTCAAGCTCCTCCTCAGC
AGCAGCGAGTGGGTGCAGTCGGAGAA
GCTGGTGCGGGCGCTGCAGACTCCGC
TGATGAGGCTGTGCGCCTGGTACCTG
TATGGAGAGAAGCACCGCGGCTACGC
GCTGAACCCCGTGGCCAACTTCCACC
TGCAGAACGGGGCGGTGCTGTGGCGC
ATCAACTGGATGGCGGATGTGAGCCT
CAGAGGCATCACCGGCTCCTGCGGCC
TGATGGCCAACTACCGCTACTTCCTG
GAGGAGACGGGCCCCAACAGCACCTC
CTACCTCGGCTCCAAGATCATCAAAG
CCTCTGAGCAGGTCCTCAGCCTAGTG
GCCCAGTTTCAAAAGAACTGACAGTA
AAGGTGGATACGGATCCGAA |
Final protein sequence (Tag sequence in lowercase):
mhhhhhhssgvdlgtenlyfq^smEL
LRRAVPPTPAYELRAATPAPAEGQCA
DFVSFYGGLAETAQRAELLGRLARGF
GVDHGQVAEQSAGVLHLRQQQREAAV
LLQAEDRLRYALVPRYRGLFHHISKL
DGGVRFLVQLRADLLEAQALKLVEGP
DVREMNGVLKGMLSEWFSPGFLNLER
VTWHSPCEVLQKISEAEAVHPVKNWM
DMKRRVGPYRRCYFFSHCSTPGEPLV
VLHVALTGDISSNIQAIVKEHPPSET
AAANKITAAIFYSISLTQQGLQGVEL
GTFLIKRVVKELQREFPHLGVFSSLS
PIPGFTKWLLGLLNSQTKEHGRNELF
TDSECKEISEITGGPINETLKLLLSS
SEWVQSEKLVRALQTPLMRLCAWYLY
GEKHRGYALNPVANFHLQNGAVLWRI
NWMADVSLRGITGSCGLMANYRYFLE
ETGPNSTSYLGSKIIKASEQVLSLVA
QFQKN
^ TEV cleavage site |
Tags and additions: Cleavable N-terminal His6-tag. |
Host: BL21 (DE3)R3-pRARE2 (Phage resistant strain).
|
Growth medium, induction protocol: The glycerol stock of host strain BL21 (DE3)-R3-pRARE2 was used to inoculate 10ml of TB (terrific Broth) supplemented with 50µg/ml kanamycin and 35µg/ml chloramphenicol. This starter culture was grown overnight at 37°C and used to inoculate 3x 1 litre culture in TB supplemented with 100µg/ml kanamycin. The culture was grown at 37°C until the OD600 reached ~2.0. After that the temperature was lowered to 18°C. Protein production was induced with 1 mM IPTG and recombinant MLYCD was expressed at that temperature overnight. The next day cells were harvested by centrifugation at 5000rpm for 20 minutes then the supernatant was discarded and pellets re-suspended in 70ml of 2x lysis buffer. Stored at -80°C.
Lysis buffer: 100 mM K-phosphate, pH 7.5; 1 M NaCl; 20% Glycerol; 1 mM TCEP; 1x Protease Inhibitors Cocktail Set VII (Calbiochem, 1/1000 dilution); and 15 units/ml Benzonase.
Extraction buffer, extraction method: Frozen pellets were thawed and supplemented with TCEP, Benzonase and protease inhibitors. Cells were lysed by sonication. Nucleic acids and cell debris were removed by adding 0.15% PEI (polyethyleneimine), then centrifuged for further 30 minutes at 17,000rpm. |
Column 1: Ni-affinity, HisTrap Crude FF, 5ml (GE Healthcare). |
Column 1 Buffers:
Lysis buffer: 100 mM K-phosphate, pH 7.5; 1 M NaCl; 20% glycerol; 0.5 mM TCEP.
Wash buffer: 50 mM K-phosphate, pH 7.5; 500 mM NaCl; 10% glycerol; 0.5 mM TCEP; 30 mM Imidazole.
Elution buffer: 50 mM K-phosphate, pH 7.5; 500 mM NaCl; 10% glycerol; 0.5 mM TCEP; 300 mM Imidazole. |
Column 1 Procedure: The clarified cell extract was loaded on the column at 4ml/min on an ÄKTA express system (GE Healthcare). The column was washed with 10 volumes of lysis buffer, 10 volumes of wash buffer, and then eluted with elution buffer at 4ml/min. The eluted peak of A280 was automatically collected. |
Column 2: Gel filtration, HiLoad 16/60 Superdex S75 prep grade, 120ml (GE Healthcare). |
Column 2 Buffer: 50 mM HEPES, pH 7.5; 500 mM NaCl; 5% glycerol; 0.5 mM TCEP. |
Column 2 Procedure: The eluted fraction from the Ni-affinity column was loaded on the gel filtration column equilibrated in GF buffer at 0.80ml/min. Eluted proteins were collected in 1.8ml fractions and analyzed on SDS-PAGE. |
Mass spectrometry characterization:
Observed mass: 52891.5Da
Expected mass: 52891.7Da
|
Protein concentration: Purified protein was concentrated to 10mg/ml using a Centricon centrifugal device with a 30kDa MW cut-off, and stored in 5 mM HEPES pH 7.5, 100 mM NaCl and 1% Glycerol at -80°C. |
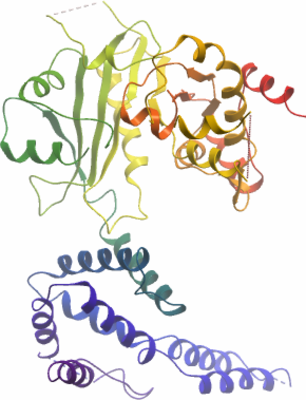
Crystallisation: Prior to crystallization, protein was pre-incubated with 2 mM decanoyl-CoA. Crystals were grown at 20°C by sitting drop vapour diffusion. A sitting drop mixing 100nl protein (10mg/ml) and 50nl well solution (10% w/v PEG20000 and 0.1 M MES pH 6.0) was equilibrated against 20µl well solution. Crystals were cryo-protected using 25% (v/v) ethylene glycol and flash cooled in liquid nitrogen. |
Data collection:
Resolution: 2.80Å.
X-ray source: Diamond Light Source beamline I02. |